Comparative Analysis of Mucor Indicus Against Aspergillus Niger and Aspergillus Fumigatus for Wheat Straw Fermentation to Produce Efficient, Inexpensive and Eco-Friendly Bioethanol
Article Information
Sana Zahra Naqvi1,2*, Syed Azhar Abbas3, Muhammad Ali-ul-Husnain Naqvi4, Nayab Batool1, Tayyaba Younas1,5*
1Institute of Microbiology, University of Agriculture, Faisalabad, Pakistan
2College of Preventive Veterinary Medicine, Nanjing Agricultural University, Nanjing, China
3School of Environment, Nanjing Normal University, Nanjing, China
4Department of Parasitology, University of Agriculture, Faisalabad, Pakistan
5Department of Chemical Engineering, Monash University, Australia
*Corresponding Author: Sana Zahra Naqvi, Institute of Microbiology, University of Agriculture, Faisalabad, Pakistan, College of Preventive Veterinary Medicine, Nanjing Agricultural University, Nanjing, China
Tayyaba Younas, Institute of Microbiology, University of Agriculture, Faisalabad, Pakistan, Department of Chemical Engineering, Monash University, Australia
Received: 27 February 2021; Accepted: 13 March 2021; Published: 19 March 2021
Citation: Sana Zahra Naqvi, Syed Azhar Abbas, Muhammad Ali-ul-Husnain Naqvi, Nayab Batool, Tayyaba Younas. Comparative Analysis of Mucor Indicus Against Aspergillus Niger and Aspergillus Fumigatus for Wheat Straw Fermentation to Produce Efficient, Inexpensive and Eco-Friendly Bioethanol. International Journal of Plant, Animal and Environmental Sciences 11 (2021): 221-232.
View / Download Pdf Share at FacebookAbstract
Increasing fuel prices and depleting hydrocarbon resources have compelled scientists to accelerate their efforts in finding reliable petroleum alternatives. Bioethanol has emerged as one of the best solutions to fit in the situation. Therefore, this colorless, biodegradable, eco-friendly liquid by all means is capable of replacing petrol. Its unique characteristics appealed modern scientists to conduct researches in this dimension more seriously. Among various production ways, bioethanol can easily be produced by fermenting lignocellulosic materials through fungus. This study compared and analyzed its production by fermenting pretreated wheat straw using three fungi; Mucor indicus, Aspergillus niger and Aspergillus fumigates separately. At first, samples collected from soil (n=111) and rotten/decomposed vegetables (n=14) were grown on yeast extract peptone dextrose (YPD) agar at 370C for 5 days. From a total of 34 isolated colonies, three most abundant fungal species were selected and identified while others were left unknown. Morphological and microscopic characterization of these fungi was done by lacto-phenol cotton blue staining. To minimize limiting factors of production, fed batch process was opted for fungal fermentation of sugars. Bioethanol was extracted by rotary evaporator while estimation was done through colorimetric method. UV induced mutagenesis enhanced ethanol production potential of these isolates. For both wild and mutant isolates, this potential was compared and analyzed graphically. At 3% sugar concentration, bioethanol production from Mucor indicus, Aspergillus niger and Aspergillus fumigatus was optimum i.e. 8.2%, 6.1% and 6.4% respectively and it became 8.4% for Mucor indicus, 6.4% for Aspergillus niger and 6.4% for Aspergillus fumigatus after 15 minutes UV exposure. Hence, under same circumstances Mucor indicus, in comparison with other two strains came out as the most competent one to produce efficient bioethanol from wheat straw ferment
Keywords
Second generation bioethanol; Wheat straw; Fermentation media; Pretreatment; Mucor indicus; Aspergillus niger; Aspergillus fumigatus
Second generation bioethanol articles; Wheat straw articles; Fermentation media articles; Pretreatment articles; Mucor indicus articles; Aspergillus niger articles; Aspergillus fumigatus articles
Article Details
Introduction
Global energy dependency has drastically increased in couple of decades. Accelerating population and modernized lifestyle are thought to be the two main factors behind [1]. Fossil fuel largely remained the source to fulfill the unceasing energy demands. At present, transportation sector alone consumes nearly 60% of the oil produced internationally, which as a consequence, contributes enormously to global air pollution, hence, jeopardizes the situation further. Its share in emitting hazardous gases like, Carbon Monoxide(CO) and Carbon Dioxide (CO2) is alarmingly high, i.e. 70% and 19% respectively. One-gallon of gasoline emits 8 kg CO2. Therefore, more vehicles moving on road means more the world is getting polluted. Taking into account 806 million vehicles in 2007 worldwide, statistic suggests that this number will surpass 1.3 billion by 2030 and 2 billion by 2050. Apart from rapid depletion of natural resources, it is clearly an environmental catastrophe which is around the corner. Consequently, it has signified the urgency of environment friendly energy alternatives. To serve the purpose, some alternates are already available for instance; bioethanol, liquefied petroleum gas (LPG), natural gas, methanol, biodiesel, boron, hydrogen, electricity, and solar fuels [2]. Among them, ‘Bioethanol’ exhibits several pluses over fossil fuels, therefore, largely known as a favorable source of renewable energy now-a-days. It is economical, ecofriendly and above all has characterized CO2-cycle in ignition [2,3]. According to the Energy Information Agency (EIA) report, there will be 60% more requirement of energy than today in 2030. If this requirement fashion of energy will prevail then world will encounter an energy crisis because universal petroleum reserves will be diminished in less than 45 years. Other important problems are global warming and environment pollution. If the normal world temperature rises further up to 20C, there will be possibly no life on earth and results into extinction of many species. So this is need of hour to produce bioethanol for wellbeing of life as prevailing place for bioethanol in fuel market [4]. Moreover, bioethanol utilizes non-edible parts of sugar cane and cereal grains like: aforesaid feed stock which is agricultural residues. Though it is the second generation of bioethanol feed stock, but it is helpful in tackling food and feed insecurity [5-7]. Researches show that wheat straw is the most probable residue in this regard, its availability and cost further supports the selection [8-11]. 1.3-1.4 kg of wheat straw are normally obtained from 1 kg wheat grain, whereas worldwide production of wheat remained nearly 680 Mt in 2011. Wheat straw; which comprises of 15% lignin, 20-30% hemicelluloses and 35−45% cellulose makes it striking feedstock for ethanol production [1].
Almost all agricultural residues produce ‘Lignocellulosic Compounds’ and these compounds could be best utilized in producing bioethanol which would have been wasted otherwise. To serve the purpose, acids are used to delignify them [12-14] then selected microbes ferment the already delignified substrate [15]. Bioconversion of various organic compounds into ethanol through fermentation is mostly carried out by fungus [7]. Lignocellulose, the main constituent of wheat straw, is an abundant resource for ethanol production and it could be hydrolyzed into a range of pentoses and hexoses like glucose, mannose, glactose, xylose and arabinose [16]. For optimum utilization of available sugars during fermentation, selection of a feasible fungus is very crucial. Similarly, features like; survival under adverse environment, substrate (moist & solid) utilization and abundant growth further elaborate the role of particular fungus selected for conversion reaction. Considering these characteristics and available data, Mucor indicus, Aspergillus niger and Aspergillus fumigatus were shortlisted to undergo initial trials for this study. Aspergillus fermentation limitation makes it less effective for bioconversion as it can only ferment a few types of hexoses for instance; glucose, however, hexoses like xylose remain unfermented [3,17]. Normally, fungi are sensitive towards high temperatures; nonetheless, Mucor is capable of fermenting pentoses and hexoses which supports its growth at temperatures where other fungi usually fail to survive. High temperature bearing potential reduces the chance of contamination as well [18]. Therefore, Mucor is economical and outstandingly suitable for fermentation in tropical regions where temperatures are normally higher [19].
Wheat straw; an abundant yet unfortunate agricultural residue which sometimes possesses zero to low commercial value has actually very high potential to be turned into a valuable bio-fuel. In present work, we have adopted simple but smart techniques to facilitate this conversion; these techniques and methodologies, due to their simple nature are easy to be implemented elsewhere. The optimum conditions discovered as a result of comparative study could be lucrative to produce bioethanol commercially. Besides, this research did not involve any laborious, expensive or hazardous activity at all.
Materials and Methods
MicroorganismsDecomposed vegetables [onion & potato (n=14)] and 111 soil samples were collected to isolate fungal strains. Collected samples were kept in labeled zip lock plastic bags until further processing. Soil samples were serially diluted and then shifted to agar medium while vegetable samples were shifted directly to the plates by tapping them over the agar plate followed by incubation at 37?C for 5 days. Samples were primarily cultured on Potato Dextrose Agar (PDA) first and then sub-cultured on Sabouraud’s (SD) and Yeast-extract Peptone Dextrose (YPD) agar. Morphological and microscopic (lacto-phenol cotton blue staining) characterization was performed to confirm the fungal specie. For lacto-phenol cotton blue staining, a drop of the stain was placed on a clean slide and a portion of the mycelia was placed on the stain using a sterilized needle and forceps. A cover slip was then placed on the wet mount and the slide was examined by a light microscope at various magnifications. The morphology and characteristics of the conidia and conidiophores were then used to classify the different types of fungus according to the standard taxonomic system [20].
Three most abundant colonies were chosen for further processing [16,21]. Confirmed species were streaked over slants of sabouraud’s dextrose agar for preservation, incubated and sealed tightly to place at 40C for further use.
Wheat Straw
Wheat straw was collected from agricultural farm of ‘University of Agriculture, Faisalabad’. To store these straws, washing, cleaning, drying, and moisture removal were carried out. Wheat straw samples were then kept in sterile zip bags.
Media preparation
Media used were PDA (potato starch-4g, dextrose-20g and agar-15g per liter of distilled water), SDA (peptone-10g, dextrose-40g and agar-15g per liter of distilled water) and YPD agar (yeast extract-10g, peptone-20g, dextrose-20g and agar-15g per liter of distilled water). Broth media, when required, were prepared following the same concentrations without adding agar. The pH was adjusted as 5.6 for PDA and SDA & for YPD pH corrected at 6.5. All the media were prepared following standard methods and autoclaved at 1210C, 15 lb pressure for 15 minutes.
Fermentation media
Fermentation media was prepared by minor modification of previously described media. The pH was adjusted at 5.5 [22]. 2% sugar medium was prepared by adding 20ml acid hydrolysate, 5g yeast extract, 7.5g ammonium sulphate, 3.5g dipotassium hydro-phosphate, 1g dihydrate calcium chloride, 0.75g hepta-hydrate, 1ml vitamin solution and 1ml trace elements in 1000ml of distilled water.
Vitamin solution was composed of 0.2g p-amino benzoic acid, 1g nicotinic acid, 1g calcium pentothenate, 1g pyridoxine, 1g thiamine, 1g biotin and 25 gram inositol in 1000ml of distilled water and trace metal solution was composed of 1g EDTA, 0.9g dihydrate calcium chloride, hepta-hydrate zinc sulphate, 0.6g hepta-hydrate ferrous sulphate, 0.2g boric acid, 0.16g dihydrate manganese chloride, 0.08g dehydrate sodium molybdenum oxide, 0.06g dihydrate cobalt chloride, 0.06g penta hydrate copper sulphate and 0.02g potassium iodide in 1000ml of distilled water. Trace element and vitamin solution was added in the media after autoclave because these were sterilized by filtration (0.22µm filter) already.
Fermentation media for 3% and 4% sugar was prepared with the similar approach as it was done previously, the only exception was the volume of acid hydolysate, 20ml was replaced by 30ml and 40ml respectively [22,23].
Pretreatment of substrate
Wheat straw (20g) was soaked in 100ml of 32% HCl and incubated at 320C for 24 hours at 150rpm. Post incubation, mixture of wheat straw and acid was diluted 10 times then heated at 1000C for 30 minutes. After filtration, solution was subjected to evaporation to get thick sugar syrup of 15 ml [24]. Sugar presence in the solution was confirmed by Benedict’s test [25] and quantitative analysis was estimated by Anthrone test [26]. This sugar syrup was used as a carbon source in fermentation media named as acid hydrolysate.
Fermentation
Fed batch fermentation method was followed. Inoculums were prepared by sterile distilled water (10ml) addition to Petri plates with fungal growth and spore counting was performed through haemocytometer. Cultivation was started with 50ml medium, inoculated with 1ml spore suspension (6 × 106 spore/ml, for purely yeast like growth) in anaerobic condition. Feeding rate was 50ml/hour for 5 hours. Medium was incubated for 370C at 150 rpm for 5 days. After completion of incubation, medium was filtered to remove fungal biomass. Fermentation was also done with 3% and 4% sugar fermentation media [23].
Product Recovery
Bioethanol from filtered solution was extracted by usage of rotary evaporator at 780C and 120 rpm [27].
Ethanol Analysis
0.115M solution of potassium dichromate (K2Cr2O7) was prepared. 11 sterilized test tubes were taken and divided into two groups, group 1st contained 9 test tubes of standard and group 2nd contained 9 test tubes of samples. Each test tube contained 2ml of 0.115M K2Cr2O7 and 9ml water.
In group 1st, 2ml of ethanol 0%, 2%, 4% s.... 16% in 9 test tubes respectively. In group 2nd, 2ml of extracted ethanol from samples was added respectively. Then all of these test tubes were heated in boiling water bath for 10 minutes for proper color generation. Optical density was measured at 600 nm [27,28].
UV Mutagenesis
Physical mutagenesis was carried out by UV light exposure. Spore suspension of each isolate was prepared by adding 1ml sterile distilled water to the Petri plates with fugal growth and shifted to sterilize test tubes. Then 100µl suspension from each test tube was taken and spread on YPD agar plates. Plates with inoculation were exposed to 290nm UV light at a distance of 20cm for 15, 20, 25 and 30 minutes and incubated at 370C for 5 days [29,30]. The survivals were marked as mutants and utilized for fermentation process for all given sugar concentrations of media.
Results
Isolation Results
Soil and vegetable samples which were earlier processed on YPD agar in order to grow M. indicus anaerobically and A. niger & A. fumigates aerobically, were confirmed after careful examination of their colony morphology and microscopic features. Key morphological and microscopic characteristics exhibited by these strains are described in Table 1 along with their images.
Table 1: Morphological and Microscopic Characteristics of M. indicus, A. niger and A. fumigatus
Acid Hydrolysis
After strong acid hydrolysis of wheat straw at low temperature, obtained sugar syrup was examined to confirm sugar presence. Appearance of brick red color upon Benedict test, confirmed the existence of high levels of sugar in the syrup whereas, Anthrone test rated its concentration as 0.56g/ml of syrup.
Ethanol Analysis Result
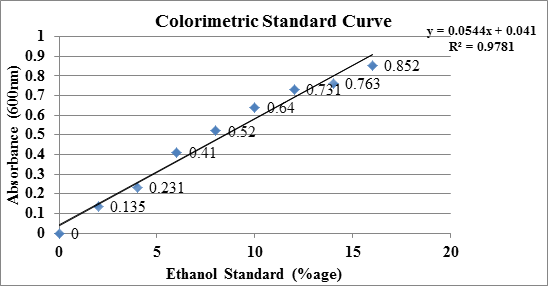
Ethanol produced after fermentation underwent quantity assessment through colorimetric method for each set. O.D. measured from Set-I of standards was plotted in a graph shown below (Figure 1).
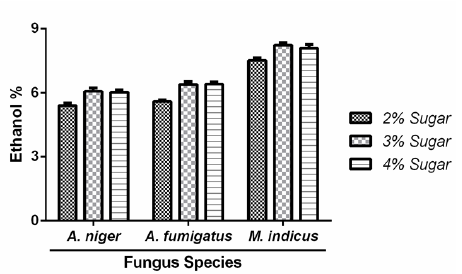
To estimate ethanol percentages, O.D of Set-II was compared with the standard curve. A graph is plotted for M. indicus, A. niger and A. fumigatus indicating ethanol percentages along with three sugar concentrations i.e. 2, 3 and 4 percent (Figure 2). 3% sugar concentration was chosen for advanced processes as it gave optimum ethanol production statistics.
Mutagenesis
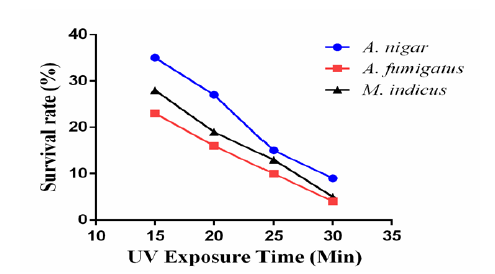
To improve ethanol production, random mutagenesis was performed by exposing M. indicus, A. niger and A. fumigatus to 290nm UV for 15, 20, 25 and 30 minutes time intervals respectively. A handsome colony number was attained at 15 minutes exposure which gradually decreased upon extra exposure (20-25 min) and turned zero at 30 minutes. Figure 3 demonstrates the survival curves of these strains.
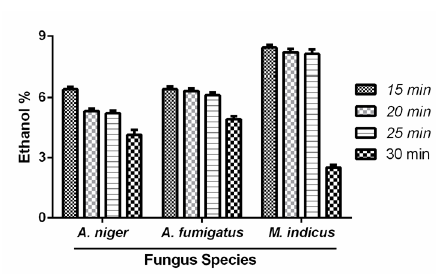
Post UV mutagenesis and fermentation of all fungal isolates with 3% sugar concentration; following results were obtained through colorimetric method (Figure 4).
Figure 1: Colorimetric method standard curve of concentration versus absorbance. The concentration of ethanol (in % age) was determined by using the equation y= 0.0544+0.041 with an R2=0.9781 where y is absorbance and x is concentration. In a standard reaction, 2ml of each ethanol concentration was mixed with 2 ml of 0.115M K2Cr2O7 reagent and heated in water bath for 10 min for color generation and then absorbance was measured at 600nm. For unknown samples (purified ethanol), 2ml of 0.115M K2Cr2O7 reagent was mixed with purified ethanol for heating and absorbance measurement. The calibration curve is needed to calculate samples concentration
Discussion
Mucorales by and large, M. indicus in particular, have been known as being amongst the most diverse and thermo-tolerant fungi [31]. Despite the fact, M. indicus has been reported to have an essential role in bioethanol production, its suitability for wheat straw fermentation has not been studied yet. Whereas, present study has revealed that M. indicus is an extremely suitable fungus for ethanol production from wheat straw. Its performance was observed well ahead of A. niger and A. fumigatus. Features like faster fermentation rate further single it out as a promising choice to produce ethanol from wheat straw. Therefore, to get faster and efficient yield, sugar from biomass was produced directly after concentrated acid pretreatment at low temperature [32]. As described in previous study, obtained sugar was fed to fermentation media along with vitamins, minerals and trace elements [23]. Like previous study, fed batch fermentation method was adopted to ensure prolonged culture lifetime and higher product recovery [8].
indicus, A. niger and A. fumigatus have long been used for ethanol production from variety of agricultural wastes [23,33,34]. While this study, in the first place, focused on their ethanol production efficiencies specifically from wheat straw. Later on, their efficiencies at different sugar concentrations (i.e. 2%, 3%, & 4%) were studied. Results of each concentration varied sharply due to the presence of limiting factors and byproducts. However, for all the three strains, optimum conversion was noted at 3% sugar fermentation media. A. niger and A. fumigatus, in this study exhibited more or less the same results for ethanol production at 3% sugar both with and without 15 minutes UV exposure. M. indicus, however, showed promising results under the same conditions. Previous studies affirmed that this is due to its capability of absolute fermentation of pentoses and hexoses into ethanol. Moreover, the greater yields along with surprising tolerance behavior against inhibitors present in hydrolysates further support the argument [16,21,35].
Although, there are numerous ethanol analysis methods like; high performance liquid chromatography [36] and gas chromatography [37,38]. However, being economical and easy to handle [27], this study applied colorimetric method. In line with the previous researches, random UV mutagenesis was attempted to enhance ethanol production [29,30]. It is noteworthy that different time intervals for UV exposure did not affect ethanol production much; consequently, our chance to study the exact pathway of mutation at molecular level was confined. Nevertheless, it opened some new avenues for future researches to engage advanced tools and mechanisms in analytic methods.
Second generation bioethanol production was previously assumed uneconomical as it involved high energy consumption during pretreatment and sugar degradation [39-42]. However, wheat straw is naturally a good culture medium for fungal growth which even at low pretreatment temperatures releases high carbohydrate content, making it a smart choice for biofuel production. Moreover, it does not involve any laborious physical pretreatments; washing, drying or vigorous milling, and hence, hydrolyzed directly by adding acid to extract sugars from cellulose [9,11]. The only requirement for proper fermentation of released sugars was; a fungal strain capable of degrading C5 and C6. Therefore, the desperate call for an effective microorganism to ferment C5 and C6 simultaneously was fulfilled comprehensively by M. indicus. Wheat straw fermentation with the help of M. indicus gave outstanding ethanol production efficiency. Moreover, their coalition at laboratory scale, turned ethanol production into a cost effective process as well. Hence, further studies are required to scale up its production at commercial levels.
Authors Contribution:
Sana Zahra Naqvi and Syed Azhar Abbas have contributed equally to this work.
References
- Del Río JC, Rencoret J, Prinsen P, et al. Structural characterization of wheat straw lignin as revealed by analytical pyrolysis, 2D-NMR, and reductive cleavage methods. Journal of Agricultural and Food Chemistry 60 (2012): 5922-5935.
- Balat M. Production of bioethanol from lignocellulosic materials via the biochemical pathway: a review. Energy Conversion and Management 52 (2011): 858-875.
- Asachi R, Karimi K, Taherzadeh MJ. Fungal autolysate as a nutrient supplement for ethanol and chitosan production by Mucor indicus. Biotechnology Letters 33 (2011): 2405-2409.
- Ahmad AL, Yasin NM, Derek CJ, et al. Microalgae as a sustainable energy source for biodiesel production: a review. Renewable and Sustainable Energy Reviews 15 (2011): 584-593.
- Gáspár M, Kálmán G, Réczey K. Corn fiber as a raw material for hemicellulose and ethanol production. Process Biochemistry 42 (2007): 1135-1139.
- Kootstra AM, Beeftink HH, Scott EL, et al. Comparison of dilute mineral and organic acid pretreatment for enzymatic hydrolysis of wheat straw. Biochemical Engineering Journal 46 (2009): 126-131.
- Talebnia F, Karakashev D, Angelidaki I. Production of bioethanol from wheat straw: an overview on pretreatment, hydrolysis and fermentation. Bioresource Technology 101 (2010): 4744-4753.
- Erdei B, Frankó B, Galbe M, et al. Glucose and xylose co-fermentation of pretreated wheat straw using mutants of S. cerevisiae TMB3400. Journal of Biotechnology 164 (2013): 50-58.
- Kaparaju P, Serrano M, Thomsen AB, et al. Bioethanol, biohydrogen and biogas production from wheat straw in a biorefinery concept. Bioresource Technology 100 (2009): 2562-2568.
- Antizar?Ladislao B, Turrion?Gomez JL. Second?generation biofuels and local bioenergy systems. Biofuels, Bioproducts and Biorefining: Innovation for a Sustainable Economy 2 (2008): 455-469.
- Linde M, Jakobsson EL, Galbe M, et al. Steam pretreatment of dilute H2SO4-impregnated wheat straw and SSF with low yeast and enzyme loadings for bioethanol production. Biomass and Bioenergy 32 (2008): 326-332.
- Esteghlalian A, Hashimoto AG, Fenske JJ, et al. Modeling and optimization of the dilute-sulfuric-acid pretreatment of corn stover, poplar and switchgrass. Bioresource Technology 59 (1997): 129-136.
- Saha BC, Iten LB, Cotta MA, et al. Dilute acid pretreatment, enzymatic saccharification and fermentation of wheat straw to ethanol. Process Biochemistry 40 (2005): 3693-3700.
- Sun Y, Cheng J. Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresource Technology 83 (2002): 1-11.
- Chandel AK, Chan ES, Rudravaram R, et al. Economics and environmental impact of bioethanol production technologies: an appraisal. Biotechnology and Molecular Biology Review 2 (2007): 14-32.
- Sues A, Millati R, Edebo L, et al. Ethanol production from hexoses, pentoses, and dilute-acid hydrolyzate by Mucor indicus. FEMS Yeast Research 5 (2005): 669-676.
- Ha SJ, Kim SR, Kim H, et al. Continuous co-fermentation of cellobiose and xylose by engineered Saccharomyces cerevisiae. Bioresource Technology 149 (2013): 525-531.
- Molaverdi M, Karimi K, Khanahmadi M, et al. Enhanced sweet sorghum stalk to ethanol by fungus Mucor indicus using solid state fermentation followed by simultaneous saccharification and fermentation. Industrial Crops and Products 49 (2013): 580-585.
- Yuangsaard N, Yongmanitchai W, Yamada M, et al. Selection and characterization of a newly isolated thermotolerant Pichia kudriavzevii strain for ethanol production at high temperature from cassava starch hydrolysate. Antonie Van Leeuwenhoek 103 (2013): 577-588.
- Mailafia S, God’spower Richard Okoh HO, Olabode K, et al. Isolation and identification of fungi associated with spoilt fruits vended in Gwagwalada market, Abuja, Nigeria. Veterinary World 10 (2017): 393.
- Lennartsson PR, Karimi K, Edebo L, et al. Effects of different growth forms of Mucor indicus on cultivation on dilute-acid lignocellulosic hydrolyzate, inhibitor tolerance, and cell wall composition. Journal of Biotechnology 143 (2009): 255-261.
- Karimi K, Brandberg T, Edebo L, et al. Fed-batch cultivation of Mucor indicus in dilute-acid lignocellulosic hydrolyzate for ethanol production. Biotechnology Letters 27 (2005): 1395-1400.
- Karimi K, Zamani A. Mucor indicus: biology and industrial application perspectives: a review. Biotechnology Advances 31 (2013): 466-481.
- Radillo JJ, Ruiz-López MA, Macías RR, et al. Fermentable sugars from lupinus rotundiflorus biomass by hydrochloric acid hydrolysis. Bioresources 6 (2011): 344-355.
- Benedict SR. A Reagent For the Detection of Reducing Sugars. J Biol Chem 5 (1908): 485-487.
- Cerning-Beroard J. A note on sugar determination by the anthrone method. Cereal Chemistry 52 (1975): 857-860.
- Das SP, Ghosh A, Gupta A, et al. Lignocellulosic fermentation of wild grass employing recombinant hydrolytic enzymes and fermentative microbes with effective bioethanol recovery. BioMed Research International 2013 (2013).
- Pourkarim F, Rahimpour E, Khoubnasabjafari M, et al. A Simple Colorimetric Method for Determination of Ethanol in Exhaled Breath Condensate. Pharmaceutical Sciences (2020).
- Taloria D, Samanta S, Das S, et al. Increase in bioethanol production by random UV mutagenesis of S. cerevisiae and by addition of zinc ions in the alcohol production media. APCBEE Procedia 2 (2012): 43-49.
- Koti S, Govumoni SP, Gentela J, et al. Enhanced bioethanol production from wheat straw hemicellulose by mutant strains of pentose fermenting organisms Pichia stipitis and Candida shehatae. SpringerPlus 5 (2016): 1-9.
- de Souza CA, Lima DX, de Oliveira RJ, et al. Mucor indicus isolated from the semiarid region of Brazil: a first record for South America. Mycotaxon 131 (2016): 897-906.
- Van Groenestijn JW, Hazewinkel JH, Bakker RR. Pre-treatment of ligno-cellulose with biological acid recycling (the Biosulfurol process). International Sugar Journal 110 (2008): 689-692.
- Karimi K, Emtiazi G, Taherzadeh MJ. Production of ethanol and mycelial biomass from rice straw hemicellulose hydrolyzate by Mucor indicus. Process Biochemistry 41 (2006): 653-658.
- Sherief AA, El-Naggar NE, Hamza SS. Bioprocessing of lignocellulosic biomass for production of bioethanol using thermotolerant Aspergillus fumigatus under solid state fermentation conditions. Biotechnology 9 (2010): 513-522.
- Abedinifar S, Karimi K, Khanahmadi M, et al. Ethanol production by Mucor indicus and Rhizopus oryzae from rice straw by separate hydrolysis and fermentation. Biomass and Bioenergy 33 (2009): 828-833.
- Taherzadeh MJ, Karimi K. Enzymatic-based hydrolysis processes for ethanol from lignocellulosic materials: A review. BioResources 2 (2007): 707-738.
- Playne MJ. Determination of ethanol, volatile fatty acids, lactic and succinic acids in fermentation liquids by gas chromatography. Journal of the Science of Food and Agriculture 36 (1985): 638-644.
- Ray NH. Gas chromatography. I. The separation and estimation of volatile organic compounds by gas?liquid partition chromatography. Journal of Applied Chemistry 4 (1954): 21-25.
- Dias MO, Junqueira TL, Cavalett O, et al. Cogeneration in integrated first and second generation ethanol from sugarcane. Chemical Engineering Research and Design 91 (2013): 1411-1417.
- Dias MO, Junqueira TL, Rossell CE, et al. Evaluation of process configurations for second generation integrated with first generation bioethanol production from sugarcane. Fuel Processing Technology 109 (2013): 84-89.
- Palacios-Bereche R, Mosqueira-Salazar KJ, Modesto M, et al. Exergetic analysis of the integrated first-and second-generation ethanol production from sugarcane. Energy 62 (2013): 46-61.
- Stephen JD, Mabee WE, Saddler JN. Will second?generation ethanol be able to compete with first?generation ethanol? Opportunities for cost reduction. Biofuels, Bioproducts and Biorefining 6 (2012): 159-176.

 Impact Factor: * 3.6
Impact Factor: * 3.6 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 