Comparison of Various Milk Samples Using Spectroscopy Chromatography, and Microscopic Analysis
Article Information
Doneti Ravinder1, Pemula Gowtham2, Karishma Khatri3, Smita C Pawar1, Mahendran Botlagunta3*
1Department of Genetics, Osmania University, Hyderabad, Telangana, India
2Department of Human Genetics and Molecular Biology, Bharathiar University, Coimbatore, India
3Department of Biotechnology, Koneru Lakshmaiah Education Foundation, Guntur, Andhra Pradesh, India
*Corresponding Author: Dr. Mahendran Botlagunta, Koneru Lakshmaiah Education Foundation, Vaddeswaram, Guntur, Andhra Pradesh, India
Received: 02 January 2021; Accepted: 26 January 2021; Published: 16 February 2021
Citation: Doneti Ravinder, Pemula Gowtham, Karishma Khatri, Smita C Pawar, Mahendran Botlagunta. Comparison of Various Milk Samples Using Spectroscopy Chromatography, and Microscopic Analysis. Journal of Food Science and Nutrition Research 4 (2021): 012-022.
View / Download Pdf Share at FacebookAbstract
Milk is one of the value-added nutritional products for animal health. Different types of milk are being used as a diet for different health benefits. Human milk also being used as trading milk for the nourishment of human health. In diary business, high value-added milk generally mixed with low-value milk for business trading. Consumption of these adulterated dairy products may cause an allergic response in certain individuals. In this study, we analyzed various milk samples, using gas chromatography (GC), Fourier-transform infrared spectroscopy (FT-IR), and Scanning Electron Microscopy (SEM) with Energy Dispersive X-Ray Analysis (EDX) to identity the difference among milk samples. FT-IR analysis showed, all milk samples show common wavelength signal at 1384 cm-1 and also distinguish various wavelength spectral patterns (human: 1077 cm-1, donkey: 1092 cm-1, buffalo: 3435 cm-1, cow: 3450 cm-1 and goat: 3443 cm-1. GC analysis results also identified common and distinct profiles for the milk samples tested. SEM-EDAX showed the presence of unique metals such as human milk contains Si-8.82wt%, donkey contains Fe-6.24wt%, and buffalo contains Si-31.29 wt% and surprisingly cow showed a very low percentage of oxygen of about 33.76 wt% when compared to other kinds of milks. Taken together, results confirmed that kinds of milks samples can be distinguished using FT-IR, GC and SEM-EDAX techniques.
Keywords
Milk samples, Adulteration, Gas Chromatography, FT-IR, SEM-EDAX
Milk samples articles; Adulteration articles; Gas Chromatography articles; FT-IR articles; SEM-EDAX articles
Milk samples articles Milk samples Research articles Milk samples review articles Milk samples PubMed articles Milk samples PubMed Central articles Milk samples 2023 articles Milk samples 2024 articles Milk samples Scopus articles Milk samples impact factor journals Milk samples Scopus journals Milk samples PubMed journals Milk samples medical journals Milk samples free journals Milk samples best journals Milk samples top journals Milk samples free medical journals Milk samples famous journals Milk samples Google Scholar indexed journals Adulteration articles Adulteration Research articles Adulteration review articles Adulteration PubMed articles Adulteration PubMed Central articles Adulteration 2023 articles Adulteration 2024 articles Adulteration Scopus articles Adulteration impact factor journals Adulteration Scopus journals Adulteration PubMed journals Adulteration medical journals Adulteration free journals Adulteration best journals Adulteration top journals Adulteration free medical journals Adulteration famous journals Adulteration Google Scholar indexed journals Gas Chromatography articles Gas Chromatography Research articles Gas Chromatography review articles Gas Chromatography PubMed articles Gas Chromatography PubMed Central articles Gas Chromatography 2023 articles Gas Chromatography 2024 articles Gas Chromatography Scopus articles Gas Chromatography impact factor journals Gas Chromatography Scopus journals Gas Chromatography PubMed journals Gas Chromatography medical journals Gas Chromatography free journals Gas Chromatography best journals Gas Chromatography top journals Gas Chromatography free medical journals Gas Chromatography famous journals Gas Chromatography Google Scholar indexed journals FT-IR articles FT-IR Research articles FT-IR review articles FT-IR PubMed articles FT-IR PubMed Central articles FT-IR 2023 articles FT-IR 2024 articles FT-IR Scopus articles FT-IR impact factor journals FT-IR Scopus journals FT-IR PubMed journals FT-IR medical journals FT-IR free journals FT-IR best journals FT-IR top journals FT-IR free medical journals FT-IR famous journals FT-IR Google Scholar indexed journals SEM-EDAX articles SEM-EDAX Research articles SEM-EDAX review articles SEM-EDAX PubMed articles SEM-EDAX PubMed Central articles SEM-EDAX 2023 articles SEM-EDAX 2024 articles SEM-EDAX Scopus articles SEM-EDAX impact factor journals SEM-EDAX Scopus journals SEM-EDAX PubMed journals SEM-EDAX medical journals SEM-EDAX free journals SEM-EDAX best journals SEM-EDAX top journals SEM-EDAX free medical journals SEM-EDAX famous journals SEM-EDAX Google Scholar indexed journals dairy products articles dairy products Research articles dairy products review articles dairy products PubMed articles dairy products PubMed Central articles dairy products 2023 articles dairy products 2024 articles dairy products Scopus articles dairy products impact factor journals dairy products Scopus journals dairy products PubMed journals dairy products medical journals dairy products free journals dairy products best journals dairy products top journals dairy products free medical journals dairy products famous journals dairy products Google Scholar indexed journals cholesterol articles cholesterol Research articles cholesterol review articles cholesterol PubMed articles cholesterol PubMed Central articles cholesterol 2023 articles cholesterol 2024 articles cholesterol Scopus articles cholesterol impact factor journals cholesterol Scopus journals cholesterol PubMed journals cholesterol medical journals cholesterol free journals cholesterol best journals cholesterol top journals cholesterol free medical journals cholesterol famous journals cholesterol Google Scholar indexed journals curd articles curd Research articles curd review articles curd PubMed articles curd PubMed Central articles curd 2023 articles curd 2024 articles curd Scopus articles curd impact factor journals curd Scopus journals curd PubMed journals curd medical journals curd free journals curd best journals curd top journals curd free medical journals curd famous journals curd Google Scholar indexed journals goat milk fat articles goat milk fat Research articles goat milk fat review articles goat milk fat PubMed articles goat milk fat PubMed Central articles goat milk fat 2023 articles goat milk fat 2024 articles goat milk fat Scopus articles goat milk fat impact factor journals goat milk fat Scopus journals goat milk fat PubMed journals goat milk fat medical journals goat milk fat free journals goat milk fat best journals goat milk fat top journals goat milk fat free medical journals goat milk fat famous journals goat milk fat Google Scholar indexed journals micronutrients articles micronutrients Research articles micronutrients review articles micronutrients PubMed articles micronutrients PubMed Central articles micronutrients 2023 articles micronutrients 2024 articles micronutrients Scopus articles micronutrients impact factor journals micronutrients Scopus journals micronutrients PubMed journals micronutrients medical journals micronutrients free journals micronutrients best journals micronutrients top journals micronutrients free medical journals micronutrients famous journals micronutrients Google Scholar indexed journals
Article Details
1. Introduction
Milk is an excellent source of most essential minerals for humans [1]. Milk and dairy products are important components of a healthy diet. The composition of milk varies from animal species, but in every case, it has a high priority in human nutrition [2]. More than 5% of the world’s milk comes from buffaloes [3]. Buffalo milk is mostly used in South Asian Countries as the largest producers of buffalo milk are from India, China and Pakistan [4]. Buffalo milk was low in cholesterol, high in fat and calories. It has high protein, Calcium, iron, vitamin and phosphorus [5]. The value of cow milk has many health benefits for human beings to improve the immune system and it also helps in the improvement of strong bones and teeth. It produces a good form of cholesterol in the body due to the presence of omega-3-fatty acids it prevents cardiovascular conditions like heart strokes. The significance of goat milk in human nutrition has so far received very little factual and academic attention despite its medical need for some people, especially infants afflicted with various ailments, including cow milk protein sensitivities [6-8]. Goat milk proteins and fats have many significant differences in their composition from the milk of other mammalian species, especially in relative proportions of the various milk proteins, fats and in their genetic polymorphisms [9-11]. Goat milk has shorter rennet coagulation time, less resistance to heat treatment, curd firmness is weaker and cheese yields are less, which might explain significant differences to cow and other milk in digestion by infants and patients, which traditionally have been explained by the homogenized nature of goat milk fat [12-14].
Human milk is believed to provide all the nutrients, essential minerals and trace elements that are required by the normal term infant growth, until weaving with few exceptions, excessive micro ids, nutrient supplies to the mother or moderate deficiencies in her diet do not greatly alter the supply to the infant. Thus, the infant is well protected by maternal homeostatic processes [15-17]. There is a wide variation in the reported data on the concentrations of trace elements in human milk from different countries. The composition of human milk varies over the course of lactation and in each individual. Human milk differs from cow’s milk both in terms of macro and micronutrients [18].
Donkey’s milk production is much more limited when compared to other species' milk [19]. It shows a closer resembles with human milk in case of protein, ash, lactose, pH and nutrition levels. It contains high levels of vitamin ‘C’, less total fat and more anti-inflammatory omega-3-fatty acids, immunity enhancing compounds particularly lysozyme and lactoferrin in helping to protect infants and peoples from disease conditions like asthma, eczema or psoriasis. It contains high volumes of vitamin B, B12 more than 60% vitamin ‘C’ when compared to that of human milk and minerals, vitamins, low volumes of fat and proteins [20-23]. In the present study, we made an attempt to investigate and compare the mineral composition of human, cow, buffalo, goat and donkey milk.
2. Materials and Methods
2.1 Materials
Milk samples from Buffalo, Cow, Goat and Donkey samples were collected from Dairy farmers from Andhra Pradesh. Human milk is collected from the local hospital of Andhra Pradesh, India, Glacial Acetic Acid, Filter papers, Funnels, Conical flasks, Beakers, Hot air oven, Water bath, Dialysis bag, Clips, Jar, Petri Dishes, Sterile Distilled Water, Magnetic Stirrer, Magnetic Beads.
2.1.1 Processing milk samples: Fresh milk samples from buffalo, cow, goat and donkey were collected. Human milk was obtained from lactating women after 3rd month of lactation to get mature milk. To the 100 mL, milk concentrated glacial acetic acid is added drop-by-drop on a magnetic stirrer, until the clear agglutination. A clear portion of the milk solution is separated from the agglutinated milk by using Whatman grade 3 chromatographic filter paper. The pH of the collected milk solution is adjusted with NaOH until it reaches to 8.0. This filtered milk solution is placed in a hot air oven at 50°C for 2 hours to reduce the volume up to 50 mL. Concentrated milk solution is subjected to centrifuge at 8000 rpm (room temperature) for 5 minutes to remove the debris.
2.1.2 Preparation of milk flakes: The collected supernatant was subjected to dialysis using a fisher brand dialysis tubing (21-152-14) with a molecular weight of 12000 to 140000 for 2 days. Immersed the dialysis membrane in a flask containing a large volume of sterile distilled water on a magnetic stirrer. Every 12 hours the water is changed. After dialysis, the sample was collected and centrifuged at 8000 rpm for 5 minutes at room temperature. The supernatant was collected without disturbing the pellet and transferred into the polystyrene Petri dishes. The plates were placed in the hot air oven at 50°C for 6-7 hours until it gets dried. A thin and transparent film/layer is observed and was collected for further biochemical characterization.
2.2 Methods
2.2.1 FTIR (Fourier Transform Infrared Spectroscopy): This technique is used to analyze the organic and inorganic compounds, both quantitatively and qualitatively. FTIR is a powerful tool that can be used for the detection of functional groups and also characterize the information of chemical bonding. FTIR uses a Fourier Transform to convert the raw data into the spectrum. FTIR spectroscopy analyses were carried out using a Jasco Fourier transform infrared spectrometer 410. FTIR spectrophotometer was connected to a photoacoustic cell in the spectral range from 450 to 4000 cm-1.
2.2.2 Gas Chromatographic (GC) Analysis: GC analysis of five milk flakes was performed using a Thermo Scientific Chemito GC 8610 system and Gas chromatograph equipped with an Elite-I, fused silica capillary column (20mm X 0.25mm ID-BP X 700-25, composed of 100% Di methyl poly siloxane). For GC detection, a Flame ionization system with ionizing energy of 70eV was used. Nitrogen gas (99.999%) was used as the carrier gas at a constant flow rate of 1mL/min and an injection volume of 5μL was employed (split ratio of 10:1); Injector temperature 100°C; Ion-source temperature 100°C. The oven temperature was programmed from 100°C (isothermal for 2 min.), with an increase of 10°C/min, to 200°C, ending with a 15 min isothermal at 200°C. Total GC running time was 30 minutes. The relative percentage amount of each component was calculated by comparing its average peak area to the total areas.
2.2.3 Energy dispersive X-ray spectroscopy: To evaluate elemental composition in five different milk flakes, we performed SEM-EDAX analysis as per the method [24-26]. Milk flakes were placed on the sample holder (stub) with carbon tape then make a conducting layer of gold-palladium with portable SC 7620 mini sputter coater or glow discharge system (Quorum Technologies Ltd.) then we load the sample in FESEM (Carl Zeiss supra 55 GEMIN German Technology) sample holding vacuum chamber. Fullscreen window and spot modes were employed depending on the size of the component. Thereafter applying the acceleration voltage of 20kv, working distance signal A in lens is a detectors and magnification of the image.
3. Results
3.1 FTIR spectroscopic Analysis
FTIR spectra of buffalo milk flakes showed absorption bands at 3435, 1641 and 1384 cm-1. Similarly, cow milk flakes displayed absorption bands at 3450, 1640 and 1384 cm-1 indicates the presence O-H, C=C, C-H as the functional groups. Goat milk flakes showed two absorption bands at 3443 and 1384 cm-1 indicates the presence of OH and CH as functional groups. Whereas in the case of human milk flakes displayed absorption bands at 3418, 1644, 1384 and 1077 cm-1. Similarly, donkey milk also showed absorption band at 3417, 1640, 1384 and 1092 cm-1 indicated the presence of OH, C=C, C-H and C=O as a functional group. Comparing these FTIR spectra we observed common functional groups in five different milk flakes are OH, C=C, CH and C=O respectively Figure 1.
3.2 Gas chromatogram analysis
Gas chromatograms of buffalo milk showed nine major peaks at retention time 3.8, 4.2, 5.3, 6.5, 8.3, 9.1, 9.9, 13.2 and 14.8 min’s. Whereas cow milk showed twelve major peaks at retention time 3.7, 4.2, 5.3, 6.5, 7.9, 8.3, 9.1, 9.9, 10.1, 10.8, 13.2 and 13.9 min’s. Similarly donkey milk twelve major peaks at various retention times 3.2, 3.8, 4.2, 5.3, 6.5, 6.8, 8.0, 8.4, 9.1, 9.9, 13.2 and 14.8 min’s. In case of human milk gas chromatograms also showed twelve peaks at different retention times 3.2, 3.6, 3.7, 4.2, 5.3, 6.5, 8.0, 8.4, 9.1, 9.3, 13.3 and 14.0 min’s respectively Figure 2. By comparing the major peaks of gas chromatograms, we identified common and unique major peaks between milk samples in the form of Venn diagram Figure 3. This diagram depicts the one major peak eluted at retention time 8.3 min’s i.e common in buffalo and cow. Two peaks eluted at retention time 9.9 and 13.2 min’s are common in buffalo, cow and donkey. Four peaks eluted at retention time 4.2, 5.3, 6.5 and 9.1 min’s is common in buffalo, cow, donkey and human. Similarly, one peak is common in cow and human and three peaks were common in donkey and human. Similarly, we noticed one unique peak at 8.3 min's in buffalo, four unique in cow, and one unique in donkey and five unique in human.
3.3 SEM-EDAX analysis
SEM image of human milk showed clear flake-like structures with 300 µm size and its corresponding EDAX spectra showed major elements are oxygen and carbon. Trace elements are Na, P, S, Cl, and Ca. Peaks possibly omitted at 6.36, 7.05, and 9.72 keV. Donkey milk showed flake-like structures with 100 µm size and its corresponding EDAX spectra showed major elements are oxygen and carbon. Trace elements are Na, P, S, Cl, and Ca. Peaks possibly omitted at 9.683 kev. Buffalo milk showed flake-like structures with 100 µm size and its corresponding EDAX spectra showed major elements are oxygen and carbon. Trace elements are Na, S, Cl, Ca, Pd and Au. Peaks possibly omitted at 1.488, 1.66, 6.418, 8.00, 9.705, 11.45 and 13.371 kev. Cow milk showed clear flake-like structures with 100 µm size and its corresponding EDAX spectra showed major elements are oxygen and carbon. Trace elements are P, S, Cl, and Ca. Peaks possibly omitted at 1.267, 8.00, 9.69, 11.48, 13.44 and 13.88 kev. Flake like structures in goat milk with 100 µm size and its corresponding EDAX spectra showed major elements are oxygen and carbon. Trace elements are Na, P, S, Cl, and Ca. Peaks possibly omitted at 9.72, 11.47 and 13.86 Kev Figure 4.
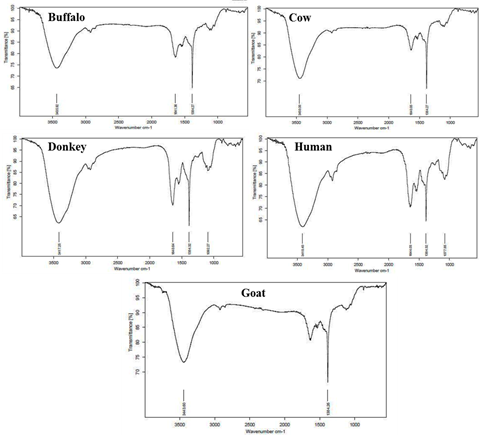
Figure 1: FTIR spectroscopy analysis of Buffalo, Cow, Donkey, Human and Goat milk samples.
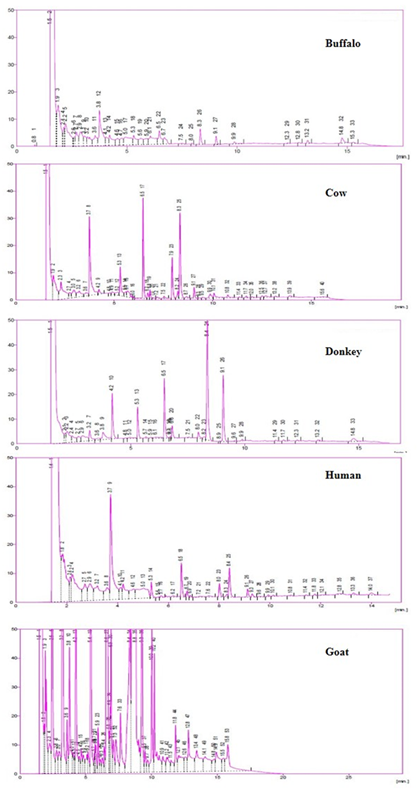
Figure 2: GC analysis of (a) Buffalo; (b) Cow; (c) Donkey; (d) Human and (e) Goat milk sample injected using conventional injector of a commercial GC system on 20mm X 0.25mm ID-BP X 700-25, composed of 100% Di methyl poly siloxane.
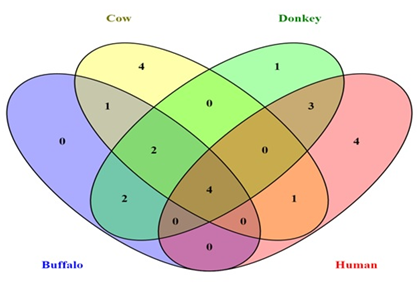
Figure 3: Venn diagram showing the number of common retention time peaks of Buffalo, Cow, Donkey and Human milk samples.
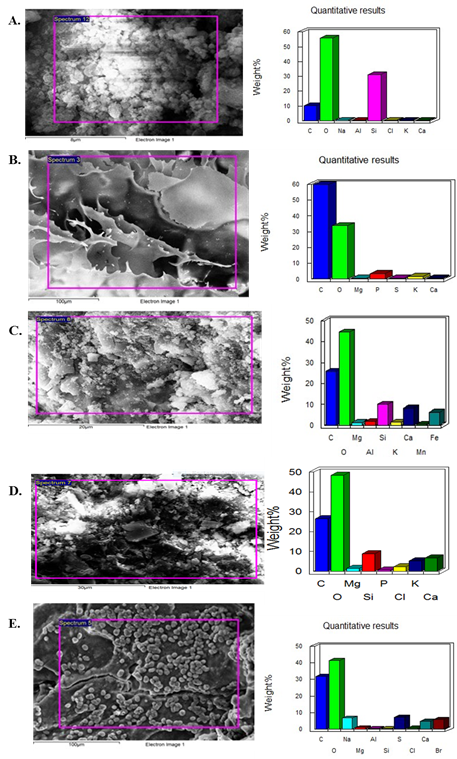
Figure 4: SEM image and Quantitative results of elemental analysis in different Milk Samples. (A) Buffalo; (B) Cow; (C) Donkey; (D) Human; (E) Goat.
4. Discussion
India occupies the first position in milk production in the world with 186 million tonnes of milk was produced in 2019, which was 22% of that year’s global total milk production of 843 million tonnes (Food and Agriculture Organization -FAO). The consumption of milk has enriched from 4.3 litre per capita per month in urban areas to 5.4 litres in last two decades, whereas in rural areas, the same improved from 3.2 litres per month to 4.3 litres per month. This growth is expected to sustain in the coming years. The increased consumption of milk and milk products, which are typically rich in protein, fat, cholesterol, macro- and microelements as well as fatty acids, is also allied with better incomes and nutritional access in developing countries [27]. Milk sources the Calcium, Vitamin D that is crucial in sustaining healthy teeth and bones, inhibit osteoporosis. Potassium increases vasodilation and drops blood pressure. Milk has induced immunoregulatory changes by increased T cell numbers in PBMC cultures of children [28]. Lactose is a sugar in milk that not all humans are able to process. The amount of lactose in milk varies differently in animals. Goat's milk, for example, has less lactose than cow's milk [29]. The enzyme, lactase digests, lactose sugar in milk and other dairy products. Some people experience decay in lactase production, those felt unpleasant and even painful, resulting in gastrointestinal discomfort upon the consumption of milk with high lactose content [30]. Even some people show allergic reaction symptoms towards the milk because their immune system reacts to milk proteins casein, alpha-lactalbumin and beta-lactalbumin as dangerous invaders [31]. Hence adulteration of milk causes potentially life-threatening for some people allergic reactions. Awkwardly milk and milk products were being very effortlessly adulterated throughout the world. Potential motives behind it may include- demand and supply gap, perishable nature of milk and lack of suitable detection tests [32]. Economic adulteration of milk was in practice by the addition of milk with a low-value ingredient, watering of milk, mixing of different species milk [33]. Need to develop analytical techniques to identify milk adulteration.
Fourier-transform infrared spectroscopy (FT-IR), Gas chromatography (GC), and Scanning Electron Microscopy (SEM) with Energy Dispersive X-Ray Analysis (EDX) to identify the type of contamination of milk with different species milk. In our study, we employed a combination of FT-IR, GC, and SEM with EDX techniques to identify the unique and common patterns among the various milk samples. Our study effectively distinguished low nutrient value milk with a high nutrient value milk sample. For example, FT-IR spectral analysis separated our samples into two distinct clusters. Donkey and human shows a similar spectral distribution pattern and display unique wavelength at 1077 and 1092 cm-1 respectively. On the other hand, buffalo, cow and goat milk samples showed a similar pattern, so this is difficult to identify individual milk type. Hence we extended our study to reveal the presence of distinct components by use of gas chromatography, GC is a widely used technique for the identification of volatile compounds. GC analysis effectively-identified various milk samples by using their unique retention time (RT). In support of the GC analysis, we further extended the study to identify the mineral content of milk samples using SEM-EDAX. All the milk samples are rich with carbon and oxygen in addition to Na, P, S, and Ca.
5. Conclusion
We can easily detect the milk adulteration by employing different techniques like Gas chromatography (GC), Fourier-transform infrared spectroscopy (FT-IR), and Scanning Electron Microscopy (SEM) with Energy Dispersive X-Ray Analysis (EDX) from the mixing of the different species milk.
Conflict of Interest
All authors declare the “No conflict of Interest”.
Acknowledgments
Doneti Ravinder was supported by UGC-BSR-RFSMS, LR.NO. F.25-1/2014-2015/BSR/No.5-78/2007 (BSR); 27/8/2015, fellowship at the Department of Genetics, Osmania University.
References
- Pietrzak-Fiecko R, Kamelska-Sadowska AM. The comparison of nutritional value of human milk with other mammals’ milk. Nutrients 12 (2020): 1404.
- Hinrichs J. Mediterranean milk and milk products. Eur. J. Nutr 43 (2004): 12-17.
- Food and Agricultural Organisation of the United Nations. The Global Dairy Sector: Factsheet. Glob. Dairy Sect. Facts (2016).
- Guo M, Hendricks G. Improving buffalo milk. In Improving the Safety and Quality of Milk: Improving Quality in Milk Products (2010): 402-416.
- Arora S, Khetra Y. Buffalo Milk Cheese. In Cheese: Chemistry, Physics and Microbiology. 4th Edn. Elsevier Inc., (2017): 1093-1101.
- Lad SS, Aparnathi KD, Mehta B, et al. Goat Milk in Human Nutrition and Health-A Review. Int. J. Curr. Microbiol. Appl. Sci 6 (2017): 1781-1792.
- Zenebe T, Ahmed N, Kabeta T, et al. Review on Medicinal and Nutritional Values of Goat Milk. Acad. J. Nutr 3 (2014): 30-39.
- Haenlein GFW. Goat milk in human nutrition. Small Ruminant Research 51 (2004): 155-163.
- Park Y. Goat Milk: Composition, Characteristics. Encyclopedia of Animal Science, 2nd (2010).
- Ceballos LS, Morales ER, La Torre Adarve G de, et al. Composition of goat and cow milk produced under similar conditions and analyzed by identical methodology. J. Food Compos. Anal 22 (2009): 322-329.
- Martin P, Bianchi L, Cebo C, et al. Genetic polymorphism of milk proteinsa. In Advanced Dairy Chemistry: Volume 1A: Proteins: Basic Aspects, 4th (2013).
- Clark S, Mora García MB. A 100-Year Review: Advances in goat milk research. J. Dairy Sci 100 (2017): 10026-10044.
- Singh H, Waungana A. Influence of heat treatment of milk on cheesemaking properties. International Dairy Journal 11 (2001): 543-551.
- Guinee TP, Gorry CB, O’Callaghan DJ, et al. The effects of composition and some processing treatments on the rennet coagulation properties of milk. Int. J. Dairy Technol 50 (1997): 99-106.
- Bates CJ, Prentice A. Breast milk as a source of vitamins, essential minerals and trace elements. Pharmacology and Therapeutics 62 (1994): 193-220.
- Hartmann PE, Sherriff JL, Mitoulas LR. Symposium: Maternal Body Composition, Caloric Restriction and Exercise during Lactation Homeostatic Mechanisms that Regulate Lactation during Energetic Stress 1,2. J. Nutr 128 (1998): 394-399.
- Donaldson SL. Life support. Health Serv J 119 (2009): 27.
- Ballard O, Morrow AL. Human Milk Composition. Nutrients and Bioactive Factors. Pediatric Clinics of North America 60 (2013): 49-74.
- Polidori P, Beghelli D, Mariani P, et al. Donkey milk production: State of the art. Ital. J. Anim. Sci 8 (2009): 677-683.
- Nayak M, Nidoni U, Hiregoudar S, et al. Composition, Characteristics, Nutritional value and Health Benefits of Donkey Milk-A Review. Dairy Science & Technology (2017).
- Gastaldi D, Bertino E, Monti G, et al. Donkey’s milk detailed lipid composition. Front. Biosci. - Elit 2 (2010): 537-546.
- Martemucci G, D’Alessandro AG. Fat content, energy value and fatty acid profile of donkey milk during lactation and implications for human nutrition. Lipids Health Dis 11 (2012): 113.
- Aspri M, Economou N, Papademas P. Donkey milk: An overview on functionality, technology, and future prospects. Food Reviews International 33 (2017): 316-333.
- Nuspl M, Wegscheider W, Angeli J, et al. Qualitative and quantitative determination of micro-inclusions by automated SEM/EDX analysis, Analytical and Bioanalytical Chemistry 379 (2004): 640-645.
- Zadora G, Brozek-Mucha Z. SEM-EDX - A useful tool for forensic examinations. Materials Chemistry and Physics 81 (2003): 345-348.
- Hamedani NF, Mahjoub AR, Khodadadi AA, et al. CeO 2 doped ZnO flower-like nanostructure sensor selective to ethanol in presence of CO and CH 4. Sensors Actuators, B Chem 169 (2012): 67-73.
- Milk Production in India. National Dairy Development Board (2017).
- Perdijk O, Splunter M van, Savelkoul HFJ, et al. Cow’s milk and immune function in the respiratory tract: Potential mechanisms. Frontiers in Immunology 9 (2018): 143.
- Goodness of Milk & Lactose Intolerance Goodness of Milk & Lactose Intolerance. GI Society (2020).
- Harrington LK, Mayberry JF. A re-appraisal of lactose intolerance. Int. J. Clin. Pract 62 (2008): 1541-1546.
- Hochwallner H, Schulmeister U, Swoboda I, et al. Cow’s milk allergy: From allergens to new forms of diagnosis, therapy and prevention. Methods 66 (2014): 22-33.
- Poonia A, Jha A, Sharma R, et al. Detection of adulteration in milk: A review. Int. J. Dairy Technol 70 (2017): 23-42.
- Kamthania M, Saxena J, Saxena K, et al. Milk Adultration?: Methods of Detection & Remedial Measures. Int. J. Eng. Tech. Res 3 (2014): 15-20.


 Impact Factor: * 3.8
Impact Factor: * 3.8 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 