Effects of Mild Impairment in Parkinson’s Disease on Balance Control
Article Information
Giovanna Machado1, Douglas Russo-Junior1, Flávia Doná2, José Barela3, Henrique Ferraz2, Sonia Silva2, Ana Barela1*
1Institute of Physical Activity and Sport Sciences, Cruzeiro do Sul University, São Paulo, Brazil
2Department of Neurology, Federal University of São Paulo, São Paulo, Brazil
3Institute of Biosciences, São Paulo State University, Rio Claro, Brazil
*Corresponding author: Ana Barela, Institute of Physical Activity and Sport Sciences, Cruzeiro do Sul University, Rua Galvão Bueno, 868, Bloco B São Paulo, 01506-000, SP, Brazil
Received: 14 October 2019; Accepted: 24 October 2019; Published: 31 October 2019
Citation: Giovanna Machado, Douglas Russo-Junior, Flávia Doná, José Barela, Henrique Ferraz, Sonia Silva, Ana Barela. Effects of mild impairment in Parkinson’s disease on balance control. Archives of Physiotherapy and Rehabilitation 2 (2019): 018-027.
View / Download Pdf Share at FacebookAbstract
Objective: The aim of this study was to examine the effects of mild impairment of PD on propulsion and balance control during gait initiation.
Design: Retrospective study.
Setting: University research laboratory.
Methods: This study included 15 individuals with PD (59.7 ± 6.8 years) and 15 individuals matched by age (59.8 ± 6.5 years) and sex with no known orthopedic or neurologic impairment that could compromise gait (control). Participants were instructed to stand quietly, looking straight ahead with their arms hanging at their sides. After a verbal command, they walked towards the end of a walkway at a comfortable speed without interruption. Center of mass (CoM) and center of pressure (CoP) in the medial-lateral (ML) and anterior-posterior (AP) directions, horizontal impulses of each limb, and the distance between CoM and CoP at specific events were obtained.
Results: Individuals with PD exhibited reduced impulse in both limbs and directions and reduced CoM-CoP distance at most events compared with controls. Although no group difference was found in the CoM trajectory length, individuals with PD exhibited shorter CoP trajectory lengths compared to controls.
Conclusion: These results suggest that PD compromises balance control, even in individuals with mild impairment, reducing the CoP trajectory length and propulsion during gait initiation. Intervention protocols for these individuals should emphasize CoP transferences to reduce postural control deficit.
Keywords
Motor Control, Stability, Balance, Propulsion, Gait Initiation
Article Details
1. Introduction
As reduced dynamic balance control contributes to incapacitation of individuals with Parkinson’s disease (PD) and leads to higher risk for falls, balance control in those individuals in the early stages of PD must be carefully investigated to develop intervention protocols to prevent accidents. A very common task utilized to investigate the dynamic balance in this population is gait initiation [1], which is the transition between a relatively stationary position and walking [2, 3]. Moreover, balance control in dynamic tasks requires a proper integration of multiple sensory systems and motor pathways to coordinate all the task components [4]. Gait initiation requires propulsion and balance control [5], and each lower limb has a specific function based on the limb that leads the task [6, 7]. For instance, because the limb that performs the first step swings forward (“swing limb”), the contralateral one (“stance limb”) must sustain the entire body weight and maintain balance over a narrower and dynamic base of support. Gait initiation can be divided in two main phases: postural and execution [8]. The postural phase comprises the period between the first dynamical phenomena [9] and the beginning of the heel-off (HO) of the swing limb, as the execution phase comprises the execution of the first step of the swing limb. Although most studies concerning PD emphasizes the postural phase [10-13], both phases must be investigated to further understand how such a complex task is achieved and performed by individuals with PD.
A method of investigating propulsion during gait initiation is through calculating the change in momentum (i.e., impulse) generated in a given direction. Because each lower limb has a specific function during gait initiation, the change in momentum of each limb must be investigated separately to better examine the possible effects of PD on propulsion. Balance control during gait initiation could be investigated through the analysis of the center of mass (CoM) and center of pressure (CoP) in the transverse plane [14]. CoM and CoP correspond, respectively, to a point equivalent to the total body mass and point location of the vertical component of the ground reaction forces (GRF) [15]. Although these two measurements are totally independent, changes in the CoP are due to the central nervous system’s response to changes in the position of the CoM. In this way, the CoP reflects the generated forces to return CoM to a balanced position. To initiate gait, the trajectory of CoM and CoP must be separated from each other [14], and the greater the distance between CoM and CoP in the transverse plane, the more active the postural control system should be [16] to maintain the upright position as the whole body is moved medial-laterally. Usually, investigations concerning gait initiation and PD have not considered the severity of the disease among participants [16-18]. The Hoehn and Yahr (HY) staging scale [19] was originally designed for describing the severity of PD [20]. Based on this scale, balance impairment emerges only in individuals with PD classified on Stage 3. However, it is important to note that the scale is attributed after an observation of the individual’s performance, which can mask possible impairments not visually present, such as measurements acquired with specific equipment that is not always available in clinical settings. In this way, investigating individuals in the early stage of PD must be considered.
Considering that both propulsion and balance maintenance are in conflict during gait initiation and that individuals with PD with mild impairment might decrease their dynamic balance capabilities, the main goal of this study was to examine the gait initiation of individuals with mild impairment in PD. More specifically, we investigated horizontal impulses and balance control during both postural and execution phases of gait initiation of individuals with PD with mild impairment in relation to age-matched controls without disability. Although individuals scaled in stage 2 of HY do not present balance impairment [19], our hypothesis was that those individuals would present different performances of gait initiation from their peers in terms of impulse and balance control.
2. Methods
2.1 Participants
We recruited 28 individuals with PD from the Movement Disorders Units of Federal University of São Paulo and Hospital do Servidor Público Estadual Francisco Morato de Oliveira. A trained examiner rated all individuals on the Motor Section of the Unified Parkinson Disease Rating Scale (UPDRS-III). The inclusion criteria were: idiopathic PD, diagnosed by an experienced specialist following the MDS clinical diagnostic criteria for PD [21]; under treatment of stable doses of levodopa; no known musculoskeletal, visual, vestibular and/or neurologic impairments other than PD; able to follow simple verbal commands; able to walk unassisted; and no surgery in the last 6 months before participation in the study, including brain deep stimulation. The exclusion criteria were: HY scale other than 2 (n=3); diagnosis of freezing of gait (n=10); and the use of any medication that could compromise the task performance. Following the inclusion and exclusion criteria, a convenience sample of 15 individuals with PD participated in this study. Fifteen individuals matched by age and sex with absence of any known orthopedic or neurologic impairment that could compromise gait and balance and no use of any medication that might affect gait and/or balance formed the control group. Table 1 presents the general characteristics of both groups. All individuals with PD were assessed during the “on” state. The Institutional Review Board approved all procedures, and all participants signed a written consent form before the experimental procedure.
|
Age (yrs.) |
Parkinson |
Control |
p-value |
|
Mean (SD) |
Mean (SD) |
||
|
60.1 (6.8) |
60.3 (6.7) |
0.935 |
|
|
Mass (kg) |
78.1 (12.4) |
73.3 (11.7) |
0.279 |
|
Height (m) |
1.63 (0.10) |
1.65 (0.08) |
0.724 |
|
Time of disease (yrs.) |
4.3 (3.4) |
- |
- |
|
UPDRS motor score |
26.4 (9.9) |
- |
- |
|
Schwab & England scale (%) |
92.7 (5.9) |
- |
- |
UPDRS: Unified Parkinson’s Disease Rating Scale
Table 1: General characteristics of individuals with Parkinson’s disease and without disability (control).
2.2 Procedures
Evaluation of gait initiation was performed on a 9-m walkway equipped with three embedded force plates (Kistler) in the middle, with two of them placed side by side, and the third one placed in front of the first two and rotated 90°. A computerized gait analysis system (VICON, Inc.) with eight infrared cameras was used to acquire data from 33 reflective markers that were placed on body landmarks based on Vicon Plug-In Full Body gait model [22]. All participants were instructed to stand quietly for approximately 3 s, with each foot on a force plate, displaced side by side, and the body weight equally distributed between both feet, looking straight ahead with their arms hanging at their sides. After a verbal command, participants walked toward the end of the walkway (approximately 4 m) at a comfortable speed without interruption as the first step touched the third force plate. The participants were barefoot, and the initial stance position was traced on the top of each force plate for consistency throughout data acquisition. A thin rubber carpet covered the entire walkway, and the participants were unaware of the displacement of each force plate. All participants practiced the task three times, and the limb that initiates gait predominantly was chosen as the leading limb. Data from force plates and cameras were sampled at 100 Hz and acquired synchronously, and a minimum of three valid trials was acquired for each participant. Trials were considered valid if the leading foot made full contact with the third force plate during the first step and if the participant walked through the walkway looking straight ahead without interruption.
2.3 Data analysis
The reconstruction of the marker trajectories and calculations of CoM were performed using Nexus software (VICON, Inc.), and all analyses were performed using a customized routine written in Matlab software (MathWorks, Inc.). Automatic identification and visual inspection to guarantee correct identification of HO [23] and toe-off (TO) of both feet and heel strike (HS) [24] of swing limb were made based on the force plate data and trajectories of reflective markers placed on the heel and second metatarsal of both feet. From force plate data, the vertical and horizontal anterior-posterior (AP) and the medial-lateral (ML) components of GRF and the trajectories of the CoP in ML and AP directions were calculated.
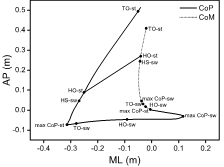
The primary outcome measures calculated for this study were impulse in the ML and AP directions for both limbs; the distance between CoM and CoP at seven specific events: HO and TO of both limbs, HS of swing limb, most lateral and most posterior displacement of CoP towards the swing limb and towards the stance limb; and length of the CoP and CoM trajectories in the ML and AP directions during postural and execution phases. The CoM-CoP distance was calculated using the Pythagorean theorem. Briefly, data from CoM and CoP in the ML and AP directions were offset at the beginning of the task, transforming all data to a common spatial origin [16]. Afterwards, the classical geometric equation (c2 = a2 + b2, where c is the distance between CoM and CoP, and a and b refer, respectively, to ML and AP directions) was employed between the coordinates of CoM and CoP at each specific event. Figure 1 shows an exemplary trajectory of CoM and CoP with the seven events, and the illustration of the distance between CoM and CoP for the HO of the stance limb. The length of CoP and CoM trajectories were calculated as the sum of the absolute values for each direction (ML and AP) and specific phase. The secondary variables were: step length and velocity of the swing limb; distance between heels before verbal command to initiate gait; anticipatory postural adjustment (APA) duration; execution duration; duration of GRF for each limb; and initial weight loading on swing and stance limbs.
Figure 1: Illustration of center of pressure (CoP) and center of mass (CoM) in the medial-lateral (ML) and anterior-posterior (AP) directions, with seven events: heel-off (HO), toe-off (TO) of swing (sw) and stance (st) limbs, heel strike (HS) of swing limb, most lateral and posterior displacement of CoP (max CoP- sw) towards the swing limb and towards the stance limb (max CoP-p- sw), and the distance between CoP and CoM at HO-st.
2.4 Statistical analysis
For all variables, the mean of three trials was obtained for each participant. One-way analysis of variance (ANOVA) and multivariate analyses of variance (MANOVA) were utilized using group (Parkinson’s and control) as a factor. The dependent variables for the ANOVAs were step length, step velocity, and distance between heels. The dependent variables for the MANOVA were impulse in the two directions for both limbs; distance between CoM and CoP at the seven events; trajectory length in the ML and AP directions for the CoP and CoM, respectively, during postural and execution phases separately; duration of APA and execution phase; GRF duration of both limbs; and initial loading on both limbs. Univariate tests were used for the MANOVA, as necessary. All analyses were performed using the Statistical Package for Social Sciences software, and an alpha level of 0.05 was adopted.
3. Results
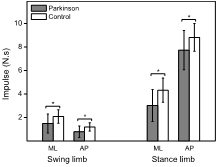
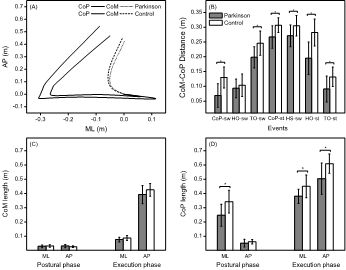
Figure 2 shows the mean (± SD) values of impulse in the ML and AP directions for both limbs. For the swing limb, individuals with PD exhibited reduced impulse in both ML (F1, 28=5·35, p=0·028) and AP (F1, 28=6·72, p=0·015) directions compared with matched controls. Similar results were found for the stance limb because individuals with PD exhibited reduced impulse in both ML (F1, 28=8·67, p=0·006) and AP (F1, 28=4·26, p=0·048) directions compared with the matched controls. Figure 3A depicts the series of the mean trajectories of CoM and CoP from all individuals of both groups in the ML and AP directions. Overall, both groups exhibited similar patterns for both trajectories, with CoP moving posteriorly and towards the swing limb and CoM moving forward and towards the stance limb. Figure 3B depicts mean (± SD) values of the distance between CoM and CoP at the seven specific events. Individuals with PD exhibited reduced CoM-CoP distance at the most lateral displacement of CoP towards the swing (F1, 28=19·46, p<0·001) and stance (F1, 28=11·48, p=0·002) limbs, TO of swing (F1, 28=11·57, p=0·002) and stance (F1, 28=8·40, p=0·007) limbs, HS of swing limb (F1, 28=6·73, p=0·015), and HO of stance limb (F1, 28=22·24, p<0·001) compared with controls. No group difference was found for HO of the swing limb (F1, 28=0·69, p=0·412). Figure 3C and 3D shows the mean (± SD) values of CoM and CoP trajectory length in the ML and AP directions during both phases. Although no group difference was found for the CoM trajectory length during postural (ML: F1, 28=0·14, p=0·707; AP: F1, 28=1·31, p=0·262) and execution (ML: F1, 28=0·57, p=0·457; AP: F1, 28=2·67, p=0·113) phases, individuals with PD exhibited shorter CoP trajectory length in the ML direction (F1, 28=10·51, p<0·003) during the postural phase, and in the ML (F1, 28=7·39, p=0·011) and AP (F1, 28=9·39, p=0·005) during the execution phase compared with controls. No group difference was found in the AP direction (F1, 28=1·55, p=0·224) during the postural phase. Table 2 shows the results of the secondary variables. Individuals with PD exhibited shorter and slower step, longer duration for both APA and execution phase, and longer duration of GRF for both limbs compared with controls. No group difference was found for distance between heels during the initial position and initial loading on both limbs.
Figure 3: Mean trajectory of center of pressure (CoP) and center of mass (CoM) in the medial-lateral (ML) and anterior-posterior (AP) directions (A); mean (± SD) values of distance between CoM and CoP at seven specific events in chronological order and (B); trajectory length of CoM (C) and CoP (D) in the ML and AP directions in the anticipatory postural adjustment (APA) and execution phases during gait initiation of individuals with Parkinson’s disease and controls without disability. Note: sw: swing limb; st: stance limb; HO: heel-off, TO: toe-off; HS: heel strike.
|
Step length (m) |
Parkinson |
Control |
F |
P-value |
|
Mean (SD) |
Mean (SD) |
|||
|
0.55 (0.09) |
0.61 (0.07) |
4.27 |
0.048 |
|
|
Step velocity (m/s) |
1.02 (0.21) |
1.18 (0.15) |
5.81 |
0.023 |
|
Distance between heels (m) |
0.15 (0.05) |
0.17 (0.04) |
1.62 |
0.214 |
|
Postural phase (APA) duration (ms) |
576 (118) |
441 (54) |
16.12 |
<0.001 |
|
Execution duration (ms) |
794 (93) |
724 (47) |
6.69 |
0.015 |
|
Ground reaction force duration (s) |
||||
|
Swing limb |
0.67 (0.12) |
0.52 (0.05) |
18.88 |
<0.001 |
|
Stance limb |
1.36 (0.15) |
1.16 (0.08) |
20.69 |
<0.001 |
|
Initial loading (% body weight) |
||||
|
Swing limb |
48.7 (6.68) |
51.4 (3.79) |
1.92 |
0.176 |
|
Stance limb |
51.3 (6.66) |
48.6 (3.80) |
1.94 |
0.175 |
APA: anticipatory postural adjustment
Table 2: Mean (SD) values of secondary variables of individuals with Parkinson’s disease and without disability (control).
4. Discussion
The goal of this study was to investigate horizontal impulses and balance control during gait initiation of individuals with PD with mild impairment. Overall, those individuals generated lower horizontal impulses under both limbs and had shorter distance between CoM-CoP at most events during gait initiation. Based on these results, our hypothesis was confirmed, indicating that individuals with PD exhibit deteriorating performance during gait initiation compared with their peers, even at the early stage of PD. Gait initiation in individuals with PD might be affected because of difficulty in generating momentum [16, 25] to move the CoM as one moves from standing still to steady-state walking. Indeed, a few attempts have inspected [26] and measured [17] forward impulse and have showed that individuals with PD exhibit reduced impulse under both the swing and stance limbs [17], which seems to further decrease in the most impaired individuals [26]. Our results, therefore, not only corroborate these scarce previous findings with a larger sample, but also clearly showed that the impulse in individuals with PD during gait initiation is also reduced in the ML direction. Reduced impulse may be due to either decreasing the force production and/or by diminishing the time interval wherein this force is produced. Interestingly, individuals with PD had a longer period of contact with the force plates, and less horizontal impulse was generated on both directions. In this way, differences in impulse production during gait initiation in individuals with PD are most likely to be due to less horizontal force production during the transition from a relatively stationary position to walking. Reduction in force production during gait initiation might be attributed to disrupted muscle activation, mainly in terms of inhibition of the soleus and prolonged activation of the tibialis anterior seen in individuals with PD [26].
Individuals with PD have shorter distance between CoM-CoP compared with individuals without PD [16, 25], and our results corroborate previous findings. Moreover, such uncoupling between CoM and CoP would indicate impairment of postural control [16]. Results from this study might shed light and aid further understanding of possible postural control changes that might be related to gait initiation as one moves from a stationary to a moving postural orientation. Our results clearly showed that reduced distance exhibited by individuals with PD was due to a shorter excursion of CoP in the ML and AP directions because no difference was found for the CoM excursion. Interestingly, the only event that the CoM-CoP distance was not different between both groups was at HO of the swing limb. At this instant, the CoP is approximately half of the distance between both the feet, as it is moving toward the stance limb, along the forward progression line. Certainly, at this instant, the person is quite stable with all forces balanced and, therefore, constituting a brief moment of “stability” that individuals with PD do not have challenged postural control. Individuals with PD have been known to have reduced limits of postural stability [27], which seems to prevent them to challenge or even to excurse the position of their CoM further from the most comfortable position of stability. Stability is even further threatened in gait initiation because of the change in the relatively stable and wide base of support provided by double-limb support to an unstable and narrower base by a single-limb support. Therefore, reducing CoP excursions, limiting to the most stable region of the base of support, is a functional and clever strategy to manage and reduce stability threatening. Differences in the performance of gait initiation in individuals with PD at stage 2 of the HY scale [19], suggesting postural control functioning impairment, indicates that the HY scale describes the clinical status and not necessarily the overall motor dysfunction. Our results showed that these individuals already show deficits in the interaction between central and peripheral commands to control balance, with disability in both phases of gait initiation. Finally, it is important to mention that the individuals with PD who participated in this study adopted a similar base of support and distributed their body weight equally between the swing and stance limbs as their peers, but their performance was still different in both the planning and execution phases.
In summary, gait initiation, due to its challenging requirements, seems to be useful to uncover possible symptoms that are not signaled by other tests and/or tasks in postural control of individuals with PD. Hence, it might constitute an important and useful instrument to evaluate and to improve postural control performance in individuals with PD. In this way, professionals and caregivers should consider strategies for improving balance control of individuals with PD with mild impairment during transition tasks, such as gait initiation. For example, Gama et al. [28] observed that the use of a body weight support system might increase the ML displacement of CoP in individuals with stroke. Similar approaches could be implemented for individuals with PD, which would provide safety and challenge the system at the same time.
5. Conclusion
PD compromises balance control, even in individuals with mild impairment, reducing CoP trajectory length and propulsion during gait initiation. These findings indicate a remarkable reduction in upright stability and its limits. Intervention protocols for these individuals should emphasize CoP transferences to reduce postural control deficit.
Competing interests
The authors declare that they have no conflict of interest.
Funding
No funding was received for this research.
Data availability
The data sets employed for analyses in the current study can be obtained upon request from the corresponding author.
References
- Delval A, Tard C, Defebvre L. Why we should study gait initiation in Parkinson's disease. Neurophysiol Clin 44 (2014): 69-76.
- Nissan M, Whittle MW. Initiation of gait in normal subjects: a preliminary study. J Biomed Eng 12 (1990): 165-171.
- Breniere Y, Do MC. When and how does steady state gait movement induced from upright posture begin? J Biomech 19 (1986): 1035-1040.
- Chang HA, Chuang TY, Lee SJ, et al. Temporal differences in relative phasing of gait initiation and first step length in patients with cervical and lumbosacral spinal cord injuries. Spinal Cord 42 (2004): 281-289.
- Polcyn AF, Lipsitz LA, Kerrigan DC, et al. Age-related changes in the initiation of gait: degradation of central mechanisms for momentum generation. Arch Phys Med Rehabil 79 (1998): 1582-1589.
- Carlsoo S. The initiation of walking. Acta Anat (Basel) 65 (1966): 1-9.
- Mann RA, Hagy JL, White V, et al. The initiation of gait. J Bone Joint Surg 61 (1979): 232-239.
- Breniere Y, Cuong Do M, Bouisset S. Are dynamic phenomena prior to stepping essential to walking? Journal of Motor Behavior 19 (1987): 62-76.
- Caderby T, Dalleau G, Leroyer P, et al. Does an additional load modify the Anticipatory Postural Adjustments in gait initiation? Gait Posture 37 (2013): 144-146.
- Burleigh-Jacobs A, Horak FB, Nutt JG, et al. Step initiation in Parkinson's disease: influence of levodopa and external sensory triggers. Mov Disord 12 (1 997): 206-215.
- Mancini M, Zampieri C, Carlson-Kuhta P, et al. Anticipatory postural adjustments prior to step initiation are hypometric in untreated Parkinson's disease: an accelerometer-based approach. Eur J Neurol 16 (2009): 1028-1034.
- Lin CC, Creath RA, Rogers MW. Variability of anticipatory postural adjustments during gait initiation in individuals with Parkinson disease. J Neurol Phys Ther 40 (2016): 40-46.
- Bonora G, Mancini M, Carpinella I, et al. Gait initiation is impaired in subjects with Parkinson's disease in the OFF state: Evidence from the analysis of the anticipatory postural adjustments through wearable inertial sensors. Gait Posture 51 (2017): 218-221.
- Jian Y, Winter DA, Ishac MG, et al. Trajectory of the body COG and COP during initiation and termination of gait. Gait Posture 1 (1993): 9-22.
- Winter DA. Human balance and posture control during standing and walking. Gait Posture 3 (1995): 193-214.
- Martin M, Shinberg M, Kuchibhatla M, et al. Gait initiation in community-dwelling adults with Parkinson disease: comparison with older and younger adults without the disease. Phys Ther 82 (2002): 566-577.
- Halliday SE, Winter DA, Frank JS, et al. The initiation of gait in young, elderly, and Parkinson's disease subjects. Gait Posture 8 (1998): 8-14.
- Hass CJ, Waddell DE, Wolf SL, et al. Gait initiation in older adults with postural instability. Clin Biomech 23 (2008): 743-753.
- Hoehn MM, Yahr MD. Parkinsonism: onset, progression and mortality. Neurology 17 (1967): 427-442.
- Goetz CG, Poewe W, Rascol O, et al. Movement Disorder Society Task Force report on the Hoehn and Yahr staging scale: status and recommendations. Mov Disord 19 (2004): 1020-1028.
- Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson's disease. Mov Disord 30 (2015): 1591-1601.
- Vicon plug-in-gait product guide- foundation notes revision 2.0 March 2010. Vicon Motion System: Vicon Motion System Limited (2010).
- Mickelborough J, van der Linden ML, Richards J, et al. Validity and reliability of a kinematic protocol for determining foot contact events. Gait Posture 11 (2000): 32-37.
- O'Connor CM, Thorpe SK, O'Malley MJ, et al. Automatic detection of gait events using kinematic data. Gait Posture 25 (2007): 469-474.
- Hass CJ, Waddell DE, Fleming RP, et al. Gait initiation and dynamic balance control in Parkinson's disease. Arch Phys Med Rehabil 86 (2005): 2172-2176.
- Crenna P, Frigo C, Giovanni P, et al. The initiation of gait in Parkinson's disease. In: Berardelli R, Benecke R, Mafredi M, Marsden CD, editors. Motor Disturbances II. San Diego, CA: Academic Press (1990): 161-73.
- van Wegen EE, van Emmerik RE, Wagenaar RC, et al. Stability boundaries and lateral postural control in parkinson's disease. Motor Control 5 (2001): 254-269.
- Gama GL, Celestino ML, Barela JA, et al. Gait initiation and partial body weight unloading for functional improvement in post-stroke individuals Gait Posture 68 (2019): 305-310.

 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 