Endocarditis Complicated by Septic Embolic Induced Acute Myocardial Infarction
Article Information
Warner Rintje Postma*, Renicus Suffridus Hermanides, George Brandon Bravo Bruinsma, Arnoud Van 't Hof
Department of Cardiology, Isala Klinieken, Dr van Heesweg 2, 8025 AB Zwolle, The Netherlands
*Corresponding Author: Warner Rintje Postma, Department of Cardiology, Isala Klinieken, Dr van Heesweg 2, 8025 AB Zwolle, The Netherlands
Received: 27 April 2020; Accepted: 06 May 2020; Published: 14 July 2020
Citation: Warner Rintje Postma, Renicus Suffridus Hermanides, George Brandon Bravo Bruinsma, Arnoud Van t Hof. Endocarditis Complicated by Septic Embolic Induced Acute Myocardial Infarction. Cardiology and Cardiovascular Medicine 4 (2020): 305-309.
View / Download Pdf Share at FacebookAbstract
A 58-year old woman with an endocarditis of the aortic valve developed an acute anterior STEMI. Manual thrombus aspiration yields an embolus with a coccoid bacterium. The patient underwent immediate surgery with an aortic bioprosthesis and a coronary artery bypass graft (CABG) with a saphenous vein to the distal LAD.
Keywords
Infective endocarditis; Coronary embolism; Acute myocardial infarction; Coronary catheterization
Article Details
Abbreviations
CAG ≡ coronary angiography
ECG ≡ electrocardiogram
ESC ≡ european society of cardiology
LAD ≡ left anterior descending artery
STEMI ≡ ST-Elevation MyocardialInfarction
TEE ≡ trans esophageal echocardiography
Case
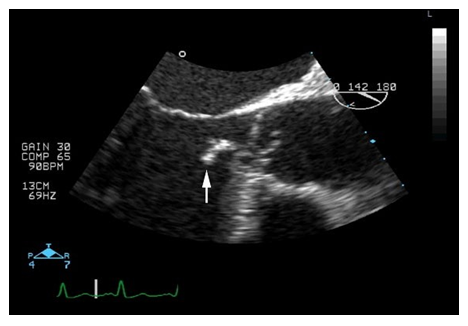
A 58-year-old woman, without any medical history of cardiovascular risk factors or other comorbidities was admitted for shortness of breath and fever since several days. A temperature of 39 degrees Celsius was measured. Cardiovascular examination revealed a high pulse pressure, and a mid-diastolic murmur at second intercostal space right. Trans esophageal echocardiography (TEE) confirmed severe aortic regurgitation due to a large vegetation (length 30 mm) attached to the left coronary cusp of the aortic valve (Figure 1).
Coronary angiography (CAG) as routine work-up for valve surgery did not show significant coronary artery disease. The patient was treated with broad spectrum antibiotics and was planned for urgent surgery, however, the evening before surgery she developed acute chest pain and ECG recording showed an acute anterior ST-Elevation Myocardial Infarction.
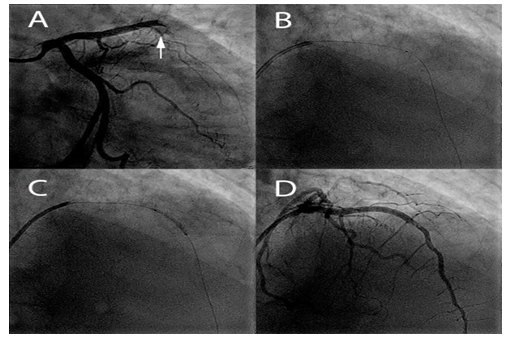
Emergent CAG demonstrated an acute occlusion of the mid left anterior descending artery (LAD) (Figure 2). Manual thrombus aspiration yields a large embolus (Figure 3).
Figure 2: Acute Coronary Angiography
(A) Acute coronary angiography showing an occlusion of the mid left anterior descending artery (LAD) (white arrowhead). (B and C) The thrombus aspiration catheter is shown and after successful aspiration dilatation with a balloon was performed. (D) Restoration of blood flow in the LAD is visible after thrombosuction.
Blood cultures were positive for enterococcus faecium. Pathologic anatomic investigation of the embolus revealed a coccoid bacterium. The patient underwent immediate surgery. An aortic bioprosthesis and, as precaution, a venous graft to the distal LAD were placed. The patient was treated with Vancomycin for 6 weeks and had an uneventful recovery afterwards. We believe that this combined approach prevented serious complications.
Discussion
Infective endocarditis refers to infection of the endocardial surface of the heart; it usually refers to infection of one or more heart valves or infection of an intracardiac device. Risk factors for infective endocarditis include cardiac factors (history of prior infective endocarditis, presence of a prosthetic valve or cardiac device, or history of valvular or congenital heart disease and noncardiac factors (intravenous drug use, indwelling intravenous catheter, immunosuppression, or a recent dental or surgical procedure). The clinical manifestations of infective endocarditis are highly variable. Infective endocarditis may present as an acute, rapidly progressive infection or as a subacute or chronic disease with low- grade fever and nonspecific symptoms. Fever is the most common symptom of infective endocarditis (up to 90 percent of cases), it is often associated with chills, anorexia, and weight loss. Cardiac murmurs are observed in approximately 85 percent of patients. Supportive signs include cutaneous manifestations such as petechiae or splinter hemorrhages.
Infective endocarditis is associated with a broad array of systemic complications; these include cardiac and neurologic complications, septic emboli, metastatic infection, and systemic immune reactions. Clinical manifestations reflecting these complications may be present at the time of initial presentation and/or may develop subsequently [1].
The risk of embolization declines after institution of appropriate antimicrobial therapy, and serious embolic events weeks after such therapy is instituted are rare [2]. In patients with left-sided infective endocarditis receiving appropriate therapy, the incidence of stroke has been decreased from 4.8 to 1.7 per 1000 patient days between the first and second week of therapy [3]. However, vegetation size >10 mm and severe mobility of the vegetation are predictors of new embolic events, even after adjustment for pathogen type, and vegetation size >15 mm is a predictor of one-year mortality [4].
In the past there have been several cases of acute myocardial infarction by septic embolization in endocarditis. To the best of our knowledge this case is the first in which thrombus aspiration was combined with a CABG for revascularization of the coronary artery. Three cases were reported where thrombus aspiration was used to restore blood flow. In two of them this was sufficient and the patient recovered well [5,6]. In the other one, thrombus aspiration was not successful and percutaneous coronary intervention (PCI) by balloon angioplasty and stenting was insufficient to improve blood flow [7]. CABG (in combination with valve surgery) was described two times before in case of myocardial infarction by septic embolization [8,9]. In the first case patient recovered uneventful, however, it was unclear which vessel was used for grafting the LAD [8]. In the second one the final outcome was not described, and a saphenous vein was used for grafting the left circumflex artery [9]. The left internal mammary artery (LIMA) is generally considered as the first choice for grafting to the LAD, while the saphenous vein is chosen as a supplementary graft given its ease of use [10]. In this case a saphenous vein was used because of the high risk of graft failure by competitive flow.
Disclosures
The authors declare that there is no conflict of interest. No funding or financial support was received.
References
- Habib G, Lancellotti P, Antunes MJ, et al. 2015 ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio- Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J 36 (2015):
- Fabri J Jr, Issa VS, Pomerantzeff PM, Grinberg M, Barretto AC, Mansur AJ. Time-related distribution, risk factors and prognostic influence of embolism in patients with left-sided infective endocarditis. Int J Cardiol 110 (2006):
- Dickerman SA, Abrutyn E, Barsic B, et al. The relationship between the initiation of antimicrobial therapy and the incidence of stroke in infective endocarditis: an analysis from the ICE Prospective Cohort Study (ICE-PCS). Am Heart J 154 (2007):
- Thuny F, Di Salvo G, Belliard O, et al. Risk of embolism and death in infective endocarditis: prognostic value of echocardiography: a prospective multicenter study. Circulation 112 (2005):
- Wojciuk J, Goode GK, More RS. Unusual presentation of endocarditis as inferior STEMI. Eur Heart J 33 (2012):
- Dekam MJ, Depta JP, Lincoff AM. A rare complication of infective endocarditis. Cleve Clin J Med 77 (2010): 296–7.
- Hibbert B, Kazmi M, Veinot JP, O'Brien ER, Glover C. Infective endocarditis presenting as ST-elevation myocardial infarction: an angiographic diagnosis. Can J Cardiol 28 (2012): 515.e15–7.
- Roxas CJ, Weekes AJ. Acute myocardial infarction caused by coronary embolism from infective endocarditis. J Emerg Med 40 (2011): 509–14.
- Okai I, Inoue K, Yamaguchi N, Makinae H, Maruyama S, Komatsu K, et al. Infective endocarditis associated with acute myocardial infarction caused by septic emboli. J Cardiol Cases 1 (2010): e28–32.
- Canver C. C. Conduit options in coronary artery bypass surgery. Chest 108 (1995): 1150– 1155.



 Impact Factor: * 3.5
Impact Factor: * 3.5 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 14.80%
Acceptance Rate: 14.80%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 