Pharmacological Chronic Heart Failure Therapy in Children. Focus on Differentiated Medical Drug Support
Article Information
Anoosh Esmaeili, Dietmar Schranz*
Pediatric Heart Center, Johann Wolfgang Goethe University Clinic Frankfurt, Germany
*Corresponding Author: Dietmar Schranz, Pediatric Heart Center, Johann Wolfgang Goethe University Clinic, Theodor-Stern-Kai 6, 60622 Frankfurt
Received: 20 July 2020; Accepted: 27 July 2020; Published: 14 August 2020
Citation: Anoosh Esmaeili, Dietmar Schranz. Pharmacological Chronic Heart Failure Therapy in Children. Focus on Differentiated Medical Drug Support. Cardiology and Cardiovascular Medicine 4 (2020): 432-442.
View / Download Pdf Share at FacebookAbstract
The etiology of pediatric heart failure is heterogenous. Missing evidence-based studies lead to symptom-based therapies. Regarding children’s life expectancy, heart regenerative strategies are required. Pathophysiological differences such as the dependence of the ventricles on the preload, the tendency towards peripheral centralization with predominantly catecholamine-controlled circulatory regulation as well as age and disease-related differences in the myocardial ß-receptor physiology require individualized precision medicine with the concept of a “responder phenotype”. First-line therapy with diuretics for chronic heart failure is not indicated. Inadequate diuretic therapy is a common reason that anti-adrenergic and renin-angiotensin-aldosterone antagonists are not or only insufficiently administered. Regarding pediatric dilated cardiomyopathy, selective ß1-blockers and tissue ACE-inhibitors are indicated primarily for their activity and side-effect profile, as well as for reasons of heart regeneration. A high-selective ß1-blocker, (bisoprolol) protects against noradrenaline-associated myocytic apoptosis and necrosis, but allows β2-receptor mediated myocardial regeneration. A reduced heart rate lowers myocardial oxygen consumption and extends the diastolic filling and coronary perfusion time. Despite the fact that the risk of cardiac fibrosis in infants is lower, the combination of bisoprolol-lisinopril-spironolactone (B-L-S) is still beneficial. Effective B-L-S doses can reduce systemic vascular resistance by ensuring myocardial perfusion pressure due to an adequate intravascular volume (preload). Almost identical daily doses of bisoprolol and lisinopril (0.1-0.2 (0.4) mg/kg/day) and 0.5-1 (- 2) mg / kg spironolactone enable a high level of compliance in the treatment by the parents or young patients. In summary, individualized heart failure therapy is the prerequisite for further regenerativ
Keywords
Heart failure; Children; Pathophysiology; Therapy; Regeneration
Heart failure articles, Children articles, Pathophysiology articles, Therapy articles, Regeneration articles
Article Details
1. Introduction
Pediatric heart failure (pHF) is a common health problem [1]. When comparing heart failure in adults, the epidemiology of pHF has been examined less systematically [2]. The incidence and prevalence of pHF differs from the global perspective. According to the WHO, congenital heart malformations, rhythm disorders, cardiomyopathies, myocardial (myocarditis) and coronary (Kawasaki) inflammation and toxic changes of the myocardium (anthracycline) are the main causes of pHF in industrialized countries [3]. The etiology of pHF varies with age, genetic disposition, and living conditions (epigenetic factors). The incidence of pHF is up to 7.4/100000, the prevalence up to 83/100000 [2]. According to pathophysiological criteria, HF is defined by an insufficient systemic blood flow at rest or during physical activity, so that the cardiac output no longer guarantees the body’s metabolic requirements. The factors that generate an indicated cardiac output of 3-4l/min/m2 at rest are based not only on the four components of myocardial contraction, preload, afterload and heart rate/rhythm, but also on cardiac synchrony, ventriculo-ventricular, atrio-ventricular and ventricular-arterial interaction. Dilated cardiomyopathy (DCM) is pathophysiologically best comparable to heart failure in adults with reduced ejection fraction (HFrEF). The annual incidence of 1.1/100000 roughly corresponds to that of non-Hodgkin’s lymphoma in children and adolescent [4], but with a significantly shorter 5-years survival time [5].
Based on our recently published article on pHF with a focus on inotropic support [6], the short review article is intended to illustrate the pediatric aspects of chronic HF therapy mainly with reduced ejection fraction.
2. Method
The review article is based on a selective literature search in PubMed, taking into account age and disease-specific cardiovascular and receptor physiology, as well as the pharmacological profile of cardiovascular drug treatment.
2.1 Therapy of pediatric heart failure with reduced ejection fraction
HFrEF is often summarized as a syndrome. However, HFrEF is whether a disease in the sense of an etiological and pathogenetic unity or a syndrome as an etiologically defined unit with unknown pathogenesis. HFrEF is a disease sequence with a heterogeneous etiology and defined pathogenesis [7]. The drug treatment of HFrEF in adults is based on large cohort studies [8]. Drug therapy for adult patients with HFrEF aims to reduce the neurohumoral response using substances from the group of ACE inhibitors, angiotensin receptor blockers, beta blockers, MRA (mineral corticoid receptor antagonist), If-channel blockers, ARNI (angiotensin receptor neprilysin Inhibitors, sacubitril / valsartan) and SGLT2 (sodium-glucose linked transporter) inhibitors. Large cohort studies are always required to influence mortality [8]. Advances in the treatment of heart failure in children with their etiological diversity and very different pathogenesis can hardly be achieved through large cohort studies. Three major HF studies therefore showed no therapeutic benefit of a) carvedilol in pediatric left ventricular dysfunction [9], b) enalapril in young patients with a "single" ventricle [10] or c) valsartan in the presence of an insufficient right ventricle in subaortic position [11]. The results of these three studies have since been used to justify the fact that drugs successfully used in adults with left ventricular HFrEF are ineffective or only questionably effective in children or in patients with systemic right ventricle [10, 11]. However, these studies also show that randomized and placebo-controlled study design per se does not yet allow evidence. Neither the number of randomized patients, the high inhomogeneity of the diseases, the clinical functional classes, nor the undifferentiated use of the medication of the patients included in the three studies over long period of time allowed evidence [12]. The valsartan study in adult patients with a right systemic ventricle is also interpreted to mean that the cardiovascular drugs remain ineffective due to cardiac morphology and genetic profile [13]. This assessment neglects that the neurohumoral response of HF is less dependent on the ventricular morphology than on its degree of dysfunction. In addition, the position of the subpulmonary or subaortic chamber is more important for drug effectiveness. The differences are due to the status of the metabolizing enzymes associated with the ventricular position and the scavenger function of the lungs [14]. In this context, it should again be noted that both the adult and pediatric therapeutic approaches are currently based mainly on blocking the neurohumoral response [5, 8].
2.2 Therapeutic consequences
The success of pediatric HF treatment, if achieved, is based on hemodynamic and cardiac “remodeling” effects. Switching from a pressure-based to a flow-based treatment, avoiding cardiac stimulation, but instead relieving the heart, led to one of the greatest success stories in the treatment of complex, congenital heart defects. The mortality rate of a Norwood operation in newborns with hypoplastic left heart syndrome (HLHS) of almost 90% in the early 1990s could be changed to a survival rate of 90% using almost the same surgical technique today [15]. The avoidance of catecholamines and the preferred use of inodilators (PDE-3 inhibitors, milrinone) was an important factor of success [16]. Evidence-based studies have not been conducted, nor have they been necessary. The decision to rethink was made based on pathophysiological considerations. The continuation of chronic treatment and relief of consequential damage of the endangered singular right ventricle have so far only been carried out in a few centers [17]. Compared to HF therapy for congenital heart defects, 70% of patients with pediatric DCM are treated with ACE inhibitors, but only around 30% with β-blockers [18]. In addition, disease-related or age-specific differentiation in therapy with β-blockers or RAAS inhibitors is almost completely absent according to their pharmacological profile is almost completely missing. Angelo-American guidelines [19] recommend ACE-inhibitors, followed by ß-blockers, in children only with restrictions (functional class I, II); on the other side, chronic diuretic therapy is recommended as a "first-line" treatment, although this is not suggested either by pathophysiology or by evidence of reduced mortality.
2.3 The therapeutic concept of the "responder" phenotype
The therapeutic "responder" phenotype in toddlers and infants can be identified acutely by its efficient neurohumoral inhibition, which can be measured by surrogate parameters such as the reduction in heart and respiratory rate and by improved feeding behavior with weight gain. In this age group, functional heart regeneration takes place in a time interval of 3-12 months [20, 21]. The prerequisite is that β-blocker and RAAS inhibitors are established according to their pharmacological profile and the age- and disease-specific molecular receptor and signaling pathways [17]. Cardiac ß-adrenergic receptors (AR) reduction is common phenomenon in all forms of HF [22]. The ß1- and ß2-AR with their different intracellular signaling pathways are affected differently. End-stage DCM in adults is characterized by isolated down-regulation of ß1-AR, while pediatric DCM is associated with the joint loss of ß1 and ß2 receptors [23]. The ischemic form (ICM) and adult patients with clinically relevant mitral valve disease or in conjunction with Fallot tetralogy also show a joint reduction in ß1- and ß2-AR [22]. The age-related differences in the behavior of beta-adrenergic receptors in DCM led Miyamoto et al. [23] on the assumption that the lack of efficiency of the carvedilol children's study [9] may also be due to the double blockade of the ß-AR receptors.
Overall, however, the importance of age- and disease-specific and thus differentiated β-blocker therapy is implied [21, 22]. The molecular data are also consistent with clinical experience [17, 24]. The highly selective ß1-receptor blocker bisoprolol has an antagonistic ß1 to ß2-AR effect in a ratio of 115, carvedilol of 5 to 1 [25]. This is of great importance for cardiac-protection and regenerative reasons, but also in relation to the side effect profile (asthmoide symptoms, bronchiolitis), especially in infants with DCM [24]. The prevention of cardiomyocytic necrosis by an efficient ß1-AR blockade can even be combined with a ß2-AR agonism, if necessary, to promote repair mechanisms [26].
In the future, ß-blockers should therefore not be used according to the generation in which they were developed, but according to the desired effectiveness. Non-specific ß-blockers like propanolol or carvedilol not only have a broader anti-arrhythmic potential than highly selective ß1-AR blockers, but due to the unspecific principle of action they can have a higher regeneration for right heart failure like that of Fallot tetralogy [27, 28] and possibly carvedilol in PAH-associated RV dysfunction [29]. In addition to age and disease-specific myocardial protection and neurohumoral interaction, all ß-blockers share a heart rate-lowering effect. The advantages lie in the reduction of oxygen consumption, especially of the myocardium, and hemodynamically in the extension of the diastolic filling time of the systemic ventricle and the coronary perfusion. The importance of an isolated heart rate reduction could also be shown in children and adolescents with ivabradine (if-channel blocker) [30]. However, like the heart rate-reducing digoxin, ivabradine has no protective ß1-AR blocker effects. Age-dependent differences in calcium-dependent signal transmission also favor the inotropic use of digoxin and calcium sensitizer (levosimendan). The inhibition of membrane-bound Na+ / K-ATPase is responsible for the positive inotropic effect of digoxin in infants. The recommendation for the use of digoxin or digitoxin as a "fourth-line" drug is based on the narrow therapeutic range with potentials of intoxication (renal failure!) or negative drug-interactions (amiodarone). Levosimendan is also more effective in children with far fewer side effects (arrhythmias) than in adults [31]. Comparable age-related differences can also be found when using PDE-3 inhibitors. In contrast to adults, milrinone leads to increased AMP and phospholamban phosphorylation within the myocardium of pediatric DCM patients. The gene expression pattern of the adenylate cyclase isoforms and the phosphodiesterase family also indicates different adaptability in pHF [32]. Therefore, PDE-3 inhibitors can be used in children with high efficacy even in long-term use (bridging to transplant) with almost no side effects [33]. Another difference between pediatric to adult hearts is the HF-associated cardiac fibrosis. Compared to adult hearts, cardiac fibrosis plays a less important role in young children [34]. The situation is similar in patients with univentricular heart failure with regard to ß-adrenergic adaptation, phosphodiesterase expression and activity and their gene profile, likewise with a lower tendency to fibrosis [35, 36]. Regarding the heart regeneration potential, not only age but also illness play an important role. Infants with HLHS and DCM differ almost fundamentally in terms of the local cardiac stem cell population and successively the endogenous regeneration potential [37]. It has been shown that infants with HLHS benefit from exogenous stem cell therapy [36], while in young DCM patients the high endogenous regenerative capacity can be used through stress induction and protective medication [21, 38, 39].
2.4 Significance of RAAS inhibitors and Neprilysin inhibition
Inhibition of the neurohumoral axis, especially of the adrenergic and renin-angiotensin-androsterone system is a recognized cornerstone of all heart failure therapy [8]. From the pediatric cardiological point of view, additional aspects are important with regard to efficiency and side effects, but also with regard parental compliance, which is particularly important for the success of chronic HF therapy. Therefore, tissue ACE inhibitors such as lisinopril or ramipril are superior to serum ACE inhibitors (captopril). Their activity and side-effect profile, their long half-life and the associated single daily dose make the drugs attractive for pHF therapy [17, 24]. Provided that not only vascular, but also myocardial angiotensin II formation must be inhibited, serum ACE-I requires significantly higher doses in order to prevent intramyocardial angiotensin II formation in a similarly efficient manner. The resulting higher bradykinin levels predispose to broncho-pulmonary side effects, especially in infants, similar to a ß2-AR blockade with unspecific ß-blockers. Due to the pathophysiology of children with HFrEF, it makes less sense to applicate ACE-I as a singular drug in contrast to a β1-AR blockade alone [40]. Endogenous catecholamine release in any symptomatic pediatric HF with a consecutive heart rate increase and peripheral vasoconstriction favors combination treatment. Long-acting β1-blockers and "tissue" ACE-I in a similar dosage combined with a mineralocorticoid receptor antagonist (MRA) in a low dose, which can also be applied only once a day, are an effective therapy with almost no side effects, but high parental acceptance. In addition, a ß1-betablocker prevents a renin rebound phenomenon of ACE-I application. The basic prerequisite for effective therapy with almost no side effects of AR- and RAAS- antagonists, however, is the exclusion of an intravascular volume deficiency. In contrast to the general questioning of the effectiveness of beta-blockers and RAAS inhibitors in the treatment of pHF [13, 19], studies in children with a neprilysin inhibitor (sacubitril) in combination with an angiotensin receptor blocker (valsartan) are currently ongoing, although this is currently neither necessary nor the theoretical risk of Alzheimer's disease is excluded, if genetically predisposed. Neprilysin is an important cerebral transmitter for preventing of plaque formation [41].
2.5 Differentiated diuretic therapy
For decades, the treatment of pediatric heart failure worldwide has mainly been based on the "tripple D" scheme (diuretics, diet = fluid reduction, digitalis,). Pediatric guidelines are likely to recommend diuretics based on the first-line recommendations in adult patients. It is precisely at this point that the pathophysiology of HFrEF in adults differs from that of young children. Most patients (infants) with DCM do not need diuretics as long as only the left or sub-aortic right ventricle is affected only systolic. Children require diuretics (symptom, tachypnea), if, in addition to systolic, a diastolic dysfunctional impairment appears or an isolated (IpcPH) or combined postcapillary pulmonary hypertension (CpcPH) with impairment of the subpulmonary ventricle. Diuretic over-treatment with intravascular volume deficiency is therefore common. Loop diuretics also accelerate the symptoms of HF-induced inadequate ADH secretion (SIADH) with free water retention and hyponatremia. Based on meanwhile extensive Japanese experience, the specific V2 (vasopressin) receptor blocker tolvaptan is the form of therapy for SIADH (hyponatremic conditions) that is also efficient in children [42]. In the case of systolic-diastolic heart failure with the resulting left atrial and pulmonary congestion, the creation of restrictive atrial communication should be reconsidered instead [43]. Diuretics can be avoided or high doses reduced. The creation of a restrictive atrial septal defect is almost indispensable for the hemodynamic evaluation of a HF-induced pulmonary hypertension before the reactivity of the pulmonary vascular bed is checked and a differential diagnosis from heart transplant to heart-lung transplantation is pending. A current therapeutic concept for pediatric heart failure with HFrEF (DCM) is summarized in Figure 1.
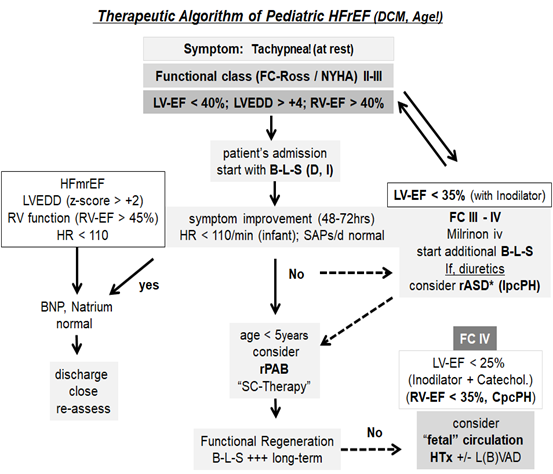
Figure 1: Children, especially infants with HFrEF (cardiac failure with reduced ejection fraction) in connection with DCM (dilated cardiomyopathy), benefit from age- and disease-specific therapy with sufficiently dosed, highly selective ß1-blockers in combination with tissue ("tissue") ACE Inhibitors and low-dose MRA (mineralocorticoid receptor antagonist), preferably spironolactone (modified according to [24]). The therapy algorithm is based on the clinical functional classes (FC); in infants and toddlers of the Ross classification. The considerations for creating a restrictive atrial communication (rASD), the age-dependent use of exogenous stem cell therapy (SC) and the use of pulmonary artery banding with the aim of left ventricular regeneration or as an alternative to the implantation of an assist device (L (B) -VAD, left or bi-ventricular assist device) for “bridging” for transplantation (HTx). Finally, the consideration of an rASD with atrial left-right shunt and an arterial right-left shunt with the establishment of a reverse Potts shunt physiology (“fetal circulation”) in the case of bi-ventricular failure with CpcPH (combined postcapillary pulmonary hypertension). Other abbreviations; B, bisoprolol; BNP, brain natriuretic peptides; Catechol, catecholamines; D, digoxin; HFmrEF, "mid-range" EF = ejection fraction; HR, heart rate; I, ivabradine; IpcPH, isolated postcapillary pulmonary hypertension; LV, left ventricle; LVEDD, left ventricular end-diastolic measurement; RV, right ventricle.
3. Main Messages
- Large cohort studies are unrealistic to demonstrate the effectiveness of HF drug treatment in children, not at least due to etiological heterogeneity of pediatric HF.
- Cardiac regeneration is inversly proportional
- to the patient’s age; drug treatment should also consider this fact.
- Only a personalized precision medicine that correspond to the molecular knowledge and the pharmacological profile of the intended drug is promising [44].
Disclosure
Both authors have no disclosures concerning the paper’s content. Dietmar Schranz is proctor for “Melody valve” Medtronic.
References
- Hsu DT, Pearson GD. Heart failure in children: part I: history, etiology, and pathophysiology. Circ Heart Fail 2 (2009): 63-70.
- Robert E Shaddy, Aneesh Thomas George, Thomas Jaecklin, Eimear Nic Lochlainn, Lalit Thakur, Rumjhum Agrawal, et al. Systematic Literature Review on the Incidence and Prevalence of Heart Failure in Children and Adolescents. Pediatr Cardiol 39 (2018): 415-436.
- Cardiac failure in children. 17th expert committee on the selection and use of essential medicines. Geneva: WHO (2008).
- Ries LG, Pollack ES, Young JL. Cancer patient survival: Surveillance, Epidemiology, and End Results Program, 1973-79. J Natl Cancer Inst 70 (1983): 693-707.
- Towbin JA, Lowe AM, Colan SD, Sleeper LA, Orav EJ, Clunie S, et al. Incidence, causes, and outcomes of dilated cardiomyopathy in children. JAMA 296 (2006): 1867-1876.
- Schranz D. Pharmacological Heart Failure Therapy in Children: Focus on Inotropic Support. Handb Exp Pharmacol 261 (2020): 177-192.
- Spranger J. Pädiatrie: Grundlagen und Praxis: Syndrome: Springer (2014).
- Berliner D, Hallbaum M, Bauersachs J. Medikamentöse Therapie der Herzinsuffizienz mit reduzierter systolischer LV-Funktion. Herz 43 (2018): 383-391.
- Robert E Shaddy , Mark M Boucek, Daphne T Hsu, Robert J Boucek, Charles E Canter, Lynn Mahony, et al. Carvedilol for children and adolescents with heart failure: a randomized controlled trial. JAMA 298 (2007): 1171-1179.
- Daphne T Hsu, Victor Zak, Lynn Mahony, Lynn A Sleeper, Andrew M Atz, Jami C Levine, et al. Enalapril in infants with single ventricle: results of a multicenter randomized trial. Circulation 122 (2010): 333-340.
- Teun van der Bom, Michiel M Winter, Berto J Bouma, Maarten Groenink, Hubert W Vliegen, Petronella G Pieper, et al. Effect of valsartan on systemic right ventricular function: a double-blind, randomized, placebo-controlled pilot trial. Circulation 127 (2013): 322-330.
- Kristin M Burns, Barry J Byrne, Bruce D Gelb, Bernhard Kühn, Leslie A Leinwand, Seema Mital, et al. New mechanistic and therapeutic targets for pediatric heart failure: report from a National Heart, Lung, and Blood Institute working group. Circulation 130 (2014): 79-86.
- Rossano JW, Shaddy RE. Update on pharmacological heart failure therapies in children: do adult medications work in children and if not, why not? Circulation 129 (2014): 607-612.
- Thum T, Borlak J. Gene expression in distinct regions of the heart. Lancet 355 (2000): 979-983.
- James S Tweddell, George M Hoffman, Kathleen A Mussatto, Raymond T Fedderly, Stuart Berger, Robert DB Jaquiss, et al. Improved survival of patients undergoing palliation of hypoplastic left heart syndrome: lessons learned from 115 consecutive patients. Circulation 106 (2002): 82-89.
- Timothy M Hoffman, Gil Wernovsky, Andrew M Atz, Thomas J Kulik, David P Nelson, Anthony C Chang, et al. Efficacy and safety of milrinone in preventing low cardiac output syndrome in infants and children after corrective surgery for congenital heart disease. Circulation 107 (2003): 996-1002.
- Schranz D, Voelkel NF. "Nihilism" of chronic heart failure therapy in children and why effective therapy is withheld. Eur J Pediatr 175 (2016): 445-455.
- Moffett BS, Price JF. National prescribing trends for heart failure medications in children. Congenit Heart Dis 10 (2015): 78-85.
- Paul F Kantor, Jane Lougheed, Adrian Dancea, Michael McGillion, Nicole Barbosa, Carol Chan, et al. Presentation, diagnosis, and medical management of heart failure in children: Canadian Cardiovascular Society guidelines. Can J Cardiol 29 (2013): 1535-1552.
- Fratz S, Hager A, Schreiber C, Schwaiger M, Hess J, Stern HC. Long-term myocardial scarring after operation for anomalous left coronary artery from the pulmonary artery. Ann Thorac Surg 92 (2011): 1761-1765.
- Schranz D, Akintuerk H, Bailey L. Pulmonary Artery Banding for Functional Regeneration of End-Stage Dilated Cardiomyopathy in Young Children: World Network Report. Circulation 137 (2018): 1410-1412.
- Brodde OE, Zerkowski HR, Borst HG, Maier W, Michel MC. Drug- and disease-induced changes of human cardiac beta 1- and beta 2-adrenoceptors. Eur Heart J 10 (1989): 38-44.
- Shelley D Miyamoto , Brian L Stauffer, Stephanie Nakano, Rebecca Sobus, Karin Nunley, Penny Nelson, et al. Beta-adrenergic adaptation in paediatric idiopathic dilated cardiomyopathy. Eur Heart J 35 (14): 33-41.
- Recla S, Schmidt D, Logeswaran T, Esmaeili A, Schranz D. Pediatric heart failure therapy: why β1-receptor blocker, tissue ACE-I and mineralocorticoid-receptor-blocker? Transl Pediatr 8 (2019): 127-132.
- Ladage D, Schwinger RHG, Brixius K. Cardio-selective beta-blocker: pharmacological evidence and their influence on exercise capacity. Cardiovasc Ther 31 (2013): 76-83.
- Manoraj Navaratnarajah, Urszula Siedlecka, Michael Ibrahim, Carin van Doorn, Gopal Soppa, Ajay Gandhi, et al. Impact of combined clenbuterol and metoprolol therapy on reverse remodelling during mechanical unloading. PLoS ONE 9 (2014): e92909.
- Honghai Liu, Cheng-Hai Zhang, Niyatie Ammanamanchi, Sangita Suresh, Christopher Lewarchik, Krithika Rao, et al. Control of cytokinesis by β-adrenergic receptors indicates an approach for regulating cardiomyocyte endowment. Sci Transl Med 11 (2019).
- Yutzey KE. Cytokinesis, Beta-Blockers, and Congenital Heart Disease. N Engl J Med 382 (2020): 291-293.
- Daniel Grinnan, Harm-Jan Bogaard, John Grizzard, Benjamin Van Tassell, Antonio Abbate, Christine DeWilde, et al. Treatment of group I pulmonary arterial hypertension with carvedilol is safe. Am J Respir Crit Care Med 189 (2014): 1562-1564.
- Bonnet D, Berger F, Jokinen E, Kantor PF, Daubeney PEF. Ivabradine in Children With Dilated Cardiomyopathy and Symptomatic Chronic Heart Failure. J Am Coll Cardiol 70 (2017): 1262-1272.
- Hummel J, Rücker G, Stiller B. Prophylactic levosimendan for the prevention of low cardiac output syndrome and mortality in paediatric patients undergoing surgery for congenital heart disease. Cochrane Database Syst Rev 8 (2017): CD011312.
- Stephanie J Nakano, Juliana Sucharov, Robert van Dusen, Mackenzie Cecil, Karin Nunley, Sean Wickers, et al. Cardiac Adenylyl Cyclase and Phosphodiesterase Expression Profiles Vary by Age, Disease, and Chronic Phosphodiesterase Inhibitor Treatment. J Card Fail 23 (201): 72-80.
- Curley M, Liebers J, Maynard R. Continuous Intravenous Milrinone Therapy in Pediatric Outpatients. J Infus Nurs 40 (2017): 92-96.
- Kathleen C Woulfe, Austine K Siomos, Hieu Nguyen, Megan SooHoo, Csaba Galambos, Brian L Stauffer, et al. Fibrosis and Fibrotic Gene Expression in Pediatric and Adult Patients with Idiopathic Dilated Cardiomyopathy. J Card Fail 23 (2017): 314-324.
- Shelley D Miyamoto, Brian L Stauffer, Jeremy Polk, Allen Medway, Matthew Friedrich, Kurt Haubold, et al. Gene expression and β-adrenergic signaling are altered in hypoplastic left heart syndrome. J Heart Lung Transplant 33 (2014): 785-793.
- Garcia AM, Beatty J-T, Nakano SJ. Heart failure in single right ventricle congenital heart disease: physiological and molecular considerations. Am J Physiol Heart Circ Physiol 318 (2020): H947-H965.
- Alexandra Traister, Rachana Patel, Anita Huang, Sarvatit Patel, Julia Plakhotnik, Jae Eun Lee, et al. Cardiac regenerative capacity is age- and disease-dependent in childhood heart disease. PLoS ONE 13 (2018): e0200342.
- Michel-Behnke I, Pavo I, Recla S, Khalil M, Jux C, Schranz D. Regenerative therapies in young hearts with structural or congenital heart disease. Transl Pediatr 8 (2019): 140-150.
- Mollova M, Bersell K, Walsh S, Savla J, Das LT, Park S-Y, et al. Cardiomyocyte proliferation contributes to heart growth in young humans. Proc Natl Acad Sci USA 110 (2013): 1446-1451.
- Buchhorn R, Hulpke-Wette M, Hilgers R, Bartmus D, Wessel A, Bürsch J. Propranolol treatment of congestive heart failure in infants with congenital heart disease: The CHF-PRO-INFANT Trial. Congestive heart failure in infants treated with propanol. Int J Cardiol 79 (2001): 167-173.
- Wesley Farris, Sonja G Schütz, John R Cirrito, Ganesh M Shankar, Xiaoyan Sun, Ana George, et al. Loss of neprilysin function promotes amyloid plaque formation and causes cerebral amyloid angiopathy. Am J Pathol 171 (2007): 241-251.
- Naoto Tominaga, Keisuke Kida, Naoki Matsumoto, Yoshihiro J Akashi, Fumihiko Miyake, Kenjiro Kimura, et al. Safety of add-on tolvaptan in patients with furosemide-resistant congestive heart failure complicated by advanced chronic kidney disease: a sub-analysis of a pharmacokinetics/ pharmacodynamics study. Clin Nephrol 84 (2015): 29-38.
- Bauer A, Esmaeili A, deRosa R, Voelkel NF, Schranz D. Restrictive atrial communication in right and left heart failure. Transl Pediatr 8 (2019): 133-139.
- Sharon Cresci, Naveen L Pereira, Ferhaan Ahmad, Mirnela Byku, Lisa de las Fuentes, David E Lanfear, et al. Heart Failure in the Era of Precision Medicine: A Scientific Statement from the American Heart Association. Circ Genom Precis Med 12 (2019): 458-485.


 Impact Factor: * 3.5
Impact Factor: * 3.5 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 14.80%
Acceptance Rate: 14.80%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 