Remote Ischemic Preconditioning in Prevention of Contrast Induced Nephropathy in Patients Undergoing Lower Limb Angioplasty Revascularization for Peripheral Arterial Disease: A Randomized Control Trial
Article Information
Debajyoti Malakar Roy1, Shrikant Digambarrao Pande2, Tar Choon Aw3 , Charyl Jia Qi Yap4, Derek John Hausenloy5, Stewart Redmond Walsh6, Julie Morris7, Tjun Yip Tang4, 8*
1Department of Renal Medicine, Changi General Hospital, Singapore
2Department of Rehabilitation Medicine, Changi General Hospital, Singapore
3Department of Biochemistry, Changi General Hospital, Singapore
4Department of Vascular Surgery, Singapore General Hospital, Singapore
5Cardiovascular & Metabolic Disorders Program, Duke-National University of Singapore Medical School, Singapore
6Department of Surgery, National University of Ireland, Galway, Ireland
7Department of Biostatistics, University of Manchester, UK
8Duke NUS Graduate Medical School, Singapore
*Corresponding Author: Tjun Yip Tang, Senior Consultant, Duke NUS Graduate Medical School, Singapore; Department of Vascular Surgery, Singapore General Hospital Level 5 Academia, 20 College Road, Singapore
Received: 06 August 2020; Accepted: 27 November 2020; Published: 04 March 2021
Citation: Debajyoti Malakar Roy, Shrikant Digambarrao Pande, Tar Choon Aw, Charyl Jia Qi Yap, Derek John Hausenloy, Stewart Redmond Walsh, Julie Morris, Tjun Yip Tang. Remote Ischemic Preconditioning in Prevention of Contrast Induced Nephropathy in Patients Undergoing Lower Limb Angioplasty Revascularization for Peripheral Arterial Disease: A Randomized Control Trial. Journal of Surgery and Research 4 (2021): 85-94.
View / Download Pdf Share at FacebookAbstract
Background: Contrast induced nephropathy (CIN) following angiography is one of the leading causes of in-hospital acute kidney injury (AKI). The aim of this study was to investigate the renoprotective effect of remote ischemic preconditioning (RIPC) to prevent CIN in patients with peripheral arterial disease (PAD) undergoing lower limb angioplasty with standard preventative measures.
Methods: 40 adult patients (eGFR >45 ml/min) undergoing peripheral arterial angioplasty received either: (1) Control: standard preventative measures comprising intravenous (IV) hydration with 0.9% normal saline (1ml/kg/hour) or (2) RIPC: four-5 min inflations and deflations of a pneumatic cuff placed on the upper arm in addition to IV hydration prior to the angiographic procedure. Serial measurements of serum creatinine, serum cystatin and urinary NGAL were taken at baseline and 2, 24, 48- and 72-hours post-procedure. The primary outcome was CIN as defined by a rise of creatinine by > 26.5umol/L above baseline within 48 hours of contrast exposure. Other outcome measures included the rise in urine NGAL and serum cystatin from base line at 2 hours, as an early marker of acute kidney injury (AKI).
Results: Both groups had similar baseline characteristics. All recruited patients had eGFR >45 ml/min, and RIPC had no renoprotective effect. AKI occurred in five (13%) patients. Changes in serum creatinine at 2 hours post-procedure did not correlate with changes in urine NGAL or serum Cystatin C.
Conclusion: In stable PAD patients (eGFR >45ml/min) undergoing lower limb angioplasty with standard preventative measures, RIPC did not offer any protection against development of CIN.
Keywords
Contrast induced nephropathy; Acute kidney injury; Remote ischemic pre-conditioning; NGAL; Cystatin-C; Peripheral arterial disease
Contrast induced nephropathy articles; Acute kidney injury articles; Remote ischemic pre-conditioning articles; NGAL articles; Cystatin-C articles; Peripheral arterial disease articles
Contrast induced nephropathy articles Contrast induced nephropathy Research articles Contrast induced nephropathy review articles Contrast induced nephropathy PubMed articles Contrast induced nephropathy PubMed Central articles Contrast induced nephropathy 2023 articles Contrast induced nephropathy 2024 articles Contrast induced nephropathy Scopus articles Contrast induced nephropathy impact factor journals Contrast induced nephropathy Scopus journals Contrast induced nephropathy PubMed journals Contrast induced nephropathy medical journals Contrast induced nephropathy free journals Contrast induced nephropathy best journals Contrast induced nephropathy top journals Contrast induced nephropathy free medical journals Contrast induced nephropathy famous journals Contrast induced nephropathy Google Scholar indexed journals Acute kidney injury articles Acute kidney injury Research articles Acute kidney injury review articles Acute kidney injury PubMed articles Acute kidney injury PubMed Central articles Acute kidney injury 2023 articles Acute kidney injury 2024 articles Acute kidney injury Scopus articles Acute kidney injury impact factor journals Acute kidney injury Scopus journals Acute kidney injury PubMed journals Acute kidney injury medical journals Acute kidney injury free journals Acute kidney injury best journals Acute kidney injury top journals Acute kidney injury free medical journals Acute kidney injury famous journals Acute kidney injury Google Scholar indexed journals Remote ischemic pre-conditioning articles Remote ischemic pre-conditioning Research articles Remote ischemic pre-conditioning review articles Remote ischemic pre-conditioning PubMed articles Remote ischemic pre-conditioning PubMed Central articles Remote ischemic pre-conditioning 2023 articles Remote ischemic pre-conditioning 2024 articles Remote ischemic pre-conditioning Scopus articles Remote ischemic pre-conditioning impact factor journals Remote ischemic pre-conditioning Scopus journals Remote ischemic pre-conditioning PubMed journals Remote ischemic pre-conditioning medical journals Remote ischemic pre-conditioning free journals Remote ischemic pre-conditioning best journals Remote ischemic pre-conditioning top journals Remote ischemic pre-conditioning free medical journals Remote ischemic pre-conditioning famous journals Remote ischemic pre-conditioning Google Scholar indexed journals Cystatin-C articles Cystatin-C Research articles Cystatin-C review articles Cystatin-C PubMed articles Cystatin-C PubMed Central articles Cystatin-C 2023 articles Cystatin-C 2024 articles Cystatin-C Scopus articles Cystatin-C impact factor journals Cystatin-C Scopus journals Cystatin-C PubMed journals Cystatin-C medical journals Cystatin-C free journals Cystatin-C best journals Cystatin-C top journals Cystatin-C free medical journals Cystatin-C famous journals Cystatin-C Google Scholar indexed journals Peripheral arterial disease articles Peripheral arterial disease Research articles Peripheral arterial disease review articles Peripheral arterial disease PubMed articles Peripheral arterial disease PubMed Central articles Peripheral arterial disease 2023 articles Peripheral arterial disease 2024 articles Peripheral arterial disease Scopus articles Peripheral arterial disease impact factor journals Peripheral arterial disease Scopus journals Peripheral arterial disease PubMed journals Peripheral arterial disease medical journals Peripheral arterial disease free journals Peripheral arterial disease best journals Peripheral arterial disease top journals Peripheral arterial disease free medical journals Peripheral arterial disease famous journals Peripheral arterial disease Google Scholar indexed journals renoprotective effect articles renoprotective effect Research articles renoprotective effect review articles renoprotective effect PubMed articles renoprotective effect PubMed Central articles renoprotective effect 2023 articles renoprotective effect 2024 articles renoprotective effect Scopus articles renoprotective effect impact factor journals renoprotective effect Scopus journals renoprotective effect PubMed journals renoprotective effect medical journals renoprotective effect free journals renoprotective effect best journals renoprotective effect top journals renoprotective effect free medical journals renoprotective effect famous journals renoprotective effect Google Scholar indexed journals therapeutic modality articles therapeutic modality Research articles therapeutic modality review articles therapeutic modality PubMed articles therapeutic modality PubMed Central articles therapeutic modality 2023 articles therapeutic modality 2024 articles therapeutic modality Scopus articles therapeutic modality impact factor journals therapeutic modality Scopus journals therapeutic modality PubMed journals therapeutic modality medical journals therapeutic modality free journals therapeutic modality best journals therapeutic modality top journals therapeutic modality free medical journals therapeutic modality famous journals therapeutic modality Google Scholar indexed journals urinary RBP articles urinary RBP Research articles urinary RBP review articles urinary RBP PubMed articles urinary RBP PubMed Central articles urinary RBP 2023 articles urinary RBP 2024 articles urinary RBP Scopus articles urinary RBP impact factor journals urinary RBP Scopus journals urinary RBP PubMed journals urinary RBP medical journals urinary RBP free journals urinary RBP best journals urinary RBP top journals urinary RBP free medical journals urinary RBP famous journals urinary RBP Google Scholar indexed journals
Article Details
1. Background
Peripheral arterial disease (PAD) affects between 3-10% of the population with prevalence rates rising with age to 15-20% in patients over 70 years old [1]. Increasing numbers of affected patients require angiography as either a diagnostic or a therapeutic modality to improve peripheral blood flow and relieve the symptoms of chronic limb ischemia (CLI). Use of iodinated contrast media during diagnostic or therapeutic procedures can lead to contrast induced nephropathy (CIN) either by direct toxic effects on tubular cells or by induction of renal ischemia [2, 3]. CIN is an important cause of hospital acquired acute kidney injury (AKI) [3]. AKI has been defined as an acute deterioration in renal function, as defined by an increase of serum creatinine by 25% or by a factor of 0.5mg/dl over baseline within 48 hours of administration of intravenous contrast and in the absence of other causes of renal dysfunction [2, 3]. Whilst the incidence of CIN in the general population is only 2%, it can be as high as 20- 30% in high-risk patients. Important risk factors for the development of CIN include pre- existing chronic kidney disease, diabetes mellitus, hypertension, elderly and congestive cardiac failure [4-6]. A large cohort study of 5787 patients with advanced PAD found that both moderate and severe renal insufficiency was associated with increased odds of death. The 1-year mortality risk was noted to be higher in patients with severe renal insufficiency FR<30ml/min/1.73m2) (OR: 2.97 95% ci: 2.39-3.69) and they tended to have a higher risk of presenting with tissue damage (ischemic ulceration or gangrene) compared with individuals having normal renal function (OR: 2.21, 95% CI: 0.64-2.98) [7]. Zaraca et al. in a recent systematic review reported on the incidence of CIN as 9.2% in patients undergoing vascular surgery. The identifiable risk factors included age >70 years, high contrast volume, pre-existing renal disease and the use of antihypertensive medications [8, 9].
Remote ischaemic preconditioning (RIPC) is a protective mechanism, which was first observed in the canine heart. Brief periods of ischemia in the limbs induced using blood pressure cuffs, have been shown to confer protection from acute kidney injury following cardiac surgery or coronary angioplasty. This protective effect has been reported to extend up to 6 months following percutaneous coronary interventions [10]. This study aims to evaluate the potential of RIPC prior to vascular angiography in reducing the risk of developing contrast-induced nephropathy. The primary end point is a reduction in the prevalence of contrast medium–induced nephropathy (defined as an increase in the serum creatinine (Ser C) concentration of >25% from the baseline value within the 72-hour period after primary angiography). Secondary outcome measures include the determination of urinary NGAL levels and cystatin C levels as early biomarkers of AKI. Additional outcome measures include length of hospital stay, need for dialysis and mortality.
2. Methods
2.1 Setting
Changi General Hospital, Singapore.
2.2 Study population
This clinical trial (NCT02516072) was in accordance with good clinical practice and the Declaration of Helsinki [11] and ethical approval was obtained from the local Institutional review board (CIRB No. 2014/2067). This study was carried out between Jan 2015 and August 2018. Eligible subjects were adult patients (>21 years of age) with PAD scheduled for peripheral angiography or angioplasty. Exclusion criteria were severe renal impairment (eGFR <30ml/min), evidence of acute renal failure or patients on dialysis, history of previous CIN, pregnancy, and contraindications to volume replacement therapy or those on Glibenclamide or Nicorandil as these may interfere with RIPC. Written informed consent was obtained from each patient who agreed to participate.
2.3 Study design
All subjects received intravenous hydration via a weight-based formula (1ml/kg/hour) with 0.9 % normal saline for 6 hours prior to the administration of contrast. A web-based service was used to generate a random sequence list with block randomization method stratified to either receive RIPC or serve as control group. The study team and statistician were blinded to treatment allocation. The control patients had a “sham” RIPC with the cuff on their arm and no inflations. Patients were randomized 1:1 to receive one of the following treatments with no intention of replacement for dropouts:
- Group 1 (Control): Patients received the intravenous hydration protocol only prior to the angiographic
- Group 2 (RIPC): Patients received the intravenous hydration protocol prior to the angiographic procedure, and RIPC, comprising placing a pneumatic cuff on the upper arm, and inflating it to 250mmHg for 5 min and deflating it for 5 min, a cycle which was repeated a total of four times. The time interval between the last cuff inflation and contrast administration were between 10-60
All patients had serial measurements of serum creatinine as well as both urinary and serum NGAL and cystatin C, at baseline prior to the procedure and 2, 24, 48- and 72-hours post- procedure.
2.4 Laboratory measurements
All analyses were performed in the Department of Laboratory Medicine at Changi General Hospital; a College of American Pathologists accredited facility. Serum creatinine was measured on the Cobas 6000 (Roche Diagnostics Asia-Pacific, Singapore) autoanalyzer using the kinetic Jaffe colorimetric assay. The analytical range of serum creatinine is from 15-2200 umol/L (0.17-24.9 mg/dl); inter-assay precision of creatinine is 2.5% at 100 umol/L (1.13 mg/dl) and 1.0% at 360 umol/L (4.07 mg/dl). Serum cystatin was also measured on the Cobas 6000 using a particle-enhanced immunoturbidimetric assay. The analytical range of serum creatinine is from 0.4-6.8 mg/L; inter-assay precision of cystatin is <2.0% at 0.92 mg/L and 1.75 mg/L. Urine NGAL was measured on the Architect i2000 analyzer (Abbott Diagnostics, Singapore) using a chemiluminescent micro particle immunoassay. The measuring range for the NGAL assay is from 10.0-1500.0 ng/mL and the inter-assay precision is <7%.
2.5 Statistical analysis
Descriptive statistics are presented as mean (SD) or median (range) for continuous variables following a Normal and non-Normal distribution respectively, and frequency and percentage for categorical variables. Fisher's Exact Test was used to assess the association between of RIPC and 25% creatinine reduction. Comparisons between the RIPC and control groups in changes in creatinine between baseline, 48 hours and 72 hours were carried out using analyses of covariance. Changes in NGAL and cystatin (which both followed non-Gaussian distributions) were made using Mann-Whitney U-test. Associations between changes in creatinine and changes in NGAL and cystatin were assessed using Spearman correlations. All comparisons were two-sided and a p value of < 0.05 was taken as statistically significant. Statistical data analysis was performed with SPSS statistical software, version 19.0.
2.6 Power calculation
The sample size calculation was based on previous studies that observed that retinol binding protein (RBP), a sensitive urinary biomarker of renal damage, peaked at 699 mg/mmol, on the second post-procedure day in the control group [12, 13]. There were no earlier studies which used rise of serum creatinine as defined by the KDIGO guidelines for diagnosis of CIN. For this study to show that RIPC would produce a 50% reduction in the urinary RBP level from 700 to 350 mg/mmol, 40 patients (half in each group) in total would be needed to give at least 80% power to show a significant difference at the 5% level.
3. Results
There were 40 subjects with a baseline eGFR > 45ml/min, these patients were randomly allocated to the control group or received RIPC in a 1:1 ratio. Of the 40 patients, 39 completed the study. One patient in the control group withdrew consent, as he did not want to undergo subsequent blood sampling (Figure 1). The treatment groups were similar with respect to demographic and co-morbidity factors except for IHD, with 74% of RIPC patients having IHD compared to 40% of the controls (Table 1). 5/39 (13%) patients developed AKI after contrast exposure, 3 in control group and 2 in the RIPC group (Fisher’s Exact test; p=1.00). No patients needed dialysis. There were no adverse events noted due to the RIPC protocol. There were no significant differences between the RIPC and control groups in the change in creatinine, NGAL or cystatin over the study period (Table 2 and Figure 2, Figure 3 and Figure 4). There was no significant association between creatinine changes and changes in NGAL and cystatin at 2 hrs, 48hrs or 72 hrs (all Spearman correlations less than 0.21).
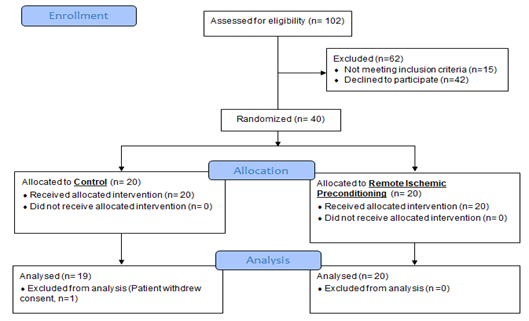
Figure 1: Consort Flowchart.
|
Mean (range) or % (number) |
|||
|
Overall cohort (n=39) |
Control (n=20) |
RIPC(n=19) |
|
|
Age (yrs.) |
65.7 (42,89) |
65.4 (52, 84) |
66.0 (42, 89) |
|
BMI |
24.7 (14.7,39.8) |
24.7 (14.7, 39.8) |
24.7 (18.1, 37.0) |
|
Systolic BP |
142 (100,194) |
136 (100, 176) |
148 (125, 194) |
|
Diastolic BP |
73.3 (50,90) |
73.5 (61, 90) |
73.2 (50, 90) |
|
DM |
90% (35) |
95% (19) |
84% (16) |
|
PVD |
100% (39) |
100% (20) |
100% (19) |
|
IHD |
56% (22) |
40% (8) |
74% (14) |
|
CVA/TIA |
23% (9) |
25% (5) |
21% (4) |
|
Hypertension |
97% (38) |
100% (20) |
95% (18) |
|
Anaemia |
5% (2) |
0% (0) |
10% (2) |
|
COPD |
5% (2) |
5% (1) |
5% (1) |
|
Malignancy |
3% (1) |
5% (1) |
0% (0) |
Table 1: Demographic factors and co-morbidities by randomised group.
|
Mean (95% CI) |
Comparison control vs RIPC |
||
|
Control |
RIPC |
||
|
Serum Creatinine |
|||
|
48 hrs. – baseline |
-2.7 (-8.7, 3.3) |
-1.0 (-7.0, 5.0) |
P=0.69 |
|
72 hrs. - baseline |
-0.4 (-7.6, 6.7) |
-0.3 (-7.5, 6.9) |
P=0.98 |
|
NGAL 1 |
|||
|
48 hrs. – baseline |
4.0 (-5.2, 28.7) |
-3.0 (-9.4, 82.7) |
P=0.71 |
|
72 hrs. - baseline |
28.8 (-13.0, 48.7) |
8.7 (-7.9, 37.4) |
P=0.96 |
|
Cystatin 1 |
|||
|
48 hrs. – baseline |
-0.06 (-0.14, 0.06) |
-0.06 (-0.24, -0.02) |
P=0.36 |
|
72 hrs. - baseline |
-0.005 (-0.12, 0.06) |
-0.01 (-0.26, 0.03) |
P=0.31 |
Table 2: Change in Serum creatinine, NGAL and Cystatin C in the RIPC and Control group between baseline and at 48 hours and 72 hours.
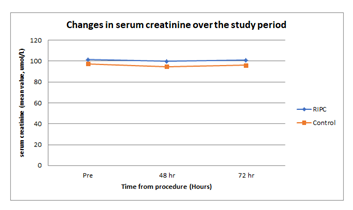
Figure 2: Changes in serum creatinine over the study period. X-axis: time (hours), Y-axis: serum creatinine (mean value, umol/L).
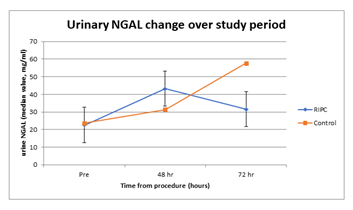
Figure 3: Urinary NGAL change over study period. X-axis: time (hours), Y-axis: urine NGAL (median value, mg/ml)
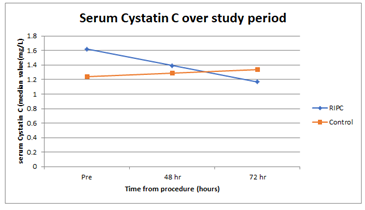
Figure 4: Serum Cystatin C over study period. X-axis: time (hours), Y-axis: serum Cystatin C (median value (mg/L)).
4. Discussion
The increasing incidence of PVD often mandates use of contrasted procedures in the form of diagnostic angiography or angioplasty. CIN has been reported in up to 9.2% of patients undergoing peripheral angiography and contributes to increased length of hospital stay, morbidity and mortality [14]. We defined CIN as an increase in serum creatinine by > 26.5 umol/L within 48 hours of contrast exposure as per the KDIGO workgroup [15]. Renal biomarkers are reported to rise earlier than serum creatinine in AKI. Serum and urine NGAL, cystatin C, urinary retinol binding protein (RBP) have all been investigated to detect AKI early [10, 16, 17]. Wang et al in a recent meta-analysis, which include 1520 patients undergoing coronary angiography found urine and plasma NGAL had a sensitivity and specificity of 84% and 89% respectively in predicting contrast induced AKI. They reported NGAL determined 4 hours after exposure to contrast was an excellent predictor of contrast- induced AKI [17]. Conversely, Moledina et al studied the performance of serum creatinine and other kidney injury biomarkers like NGAL, in a group of deceased donors who had undergone kidney biopsy at the time of organ procurement. 155/581 (27%) deceased donors had histologically proven ATN. They reported NGAL lacked accuracy in the diagnosis of acute tubular injury with an area under the receiver operating characteristic curve (AUROC) for diagnosing acute tubular injury of 0.60 (95% CI, 0.55-0.66; P=0.005) [18]. Rise in serum creatinine post-contrast exposure remains the current gold standard in the diagnosis of CIN. We did not find any correlation between rise of NGAL or Cystatin C to serum creatinine (Table 2). Preventative strategies for CIN include hydration with saline, isotonic bicarbonate; N- Acetylcysteine, RIPC and Xanthine have been reported earlier. There is still discrepancy in literature as to which preventative strategy is best [19]. RIPC has been demonstrated to have a protective effect against AKI in those undergoing primarily cardiac procedures like surgery or coronary interventions [20]. A prior study evaluating the protective effects of RIPC in patients undergoing contrasted abdominal CT scan reported a 65% risk reduction in rise of serum creatinine in the group subjected to ischemic pre-conditioning [21]. Menting et al studied a group of 76 patients at risk for development of CIN as per the Dutch guidelines. The inclusion criteria were, reduced eGFR and two additional risk factors like age, heart failure, use of diuretics etc. in those undergoing contrasted studies for diagnostic or interventional purposes [22]. While they found no difference in the change of serum creatinine from baseline to 48-72 hours, in sub-analysis of the group with a high Mehran score (>11) the rise in serum creatinine was statistically lower [22]. However, there is data, which also suggests protective measures against CIN may be ineffective. A recent network metanalysis reported on 124 trials with 28,240 patients undergoing coronary angiography, did not find either intravenous saline, isotonic bicarbonate, NGAL, xanthines or RIPC to be protective [23, 24]. From our study of 40 patients, we did not find a significant difference in creatinine, NGAL or cystatin changes between those with and without RIPC at 48- and 72-hours post contrast exposure, and no significant correlation with change in creatinine, NGAL or cystatin from pre- op to 2 hours. In our study population, 5 patients developed CIN, 3 from the control group and 2 from the RIPC group. The major strengths of this study are related to its design as a blinded study with a previously registered protocol. The physicians and nursing staff were blinded to the allocation minimizing risk of bias because of adherence to preventative measures like saline infusion and the volume of contrast used. Our study has several limitations, which may account for our findings:
- Our sample size was based on earlier studies, which investigated urinary biomarkers to detect AKI and was not based on rise in creatinine as defined by KDIGO. Hence, our sample size may have been an This is a common problem when biomarkers are used as a surrogate marker for AKI.
- RIPC was added on to other protective measures like normal saline infusion and use of low osmolar contrast media. These preventative measures may have masked the development of
- Our patient cohort had eGFR>45 ml/min. This group is less susceptible to CIN as compared to those with lower Our results may therefore represent a significant type 2 error.
The reason for inability to enroll patients with eGFR 30 to <45ml/min as per our protocol is based on our local experience, as patients chose against contrast procedures when explained the risk of CIN. We also tend to use carbon dioxide as mode of contrast under IR guidance for these patients. Authors who have found protective effect of RIPC have had majority of patients with eGFR of <45 ml/min. However, in our study, we did not find protective effect of RIPC in patients with eGFR of >45 ml/min.
5. Conclusion
We found RIPC did not offer any protective effect over and beyond saline hydration and use of low osmolar contrast in patients with eGFR>45 ml/min. The rise in Urinary NGAL and Serum Cystatin C did not correlate with rise in serum creatinine at 2 hours post contrast exposure. Since this study did not explore the possible benefits of RIPC in high-risk group (eGFR < 45 ml/min), further studies should be pursued to look at RIPC in the high-risk group of developing CIN.
Declaration
- Ethics approval and consent to participate: Singhealth IRB (CIRB NO: 2014/2067). Written informed consent.
- Consent for publication: not applicable.
- Availability of data and material: the dataset used and analysed during the current study are available from the corresponding author on reasonable request.
- Competing interest: all the authors declare that they have no competing interest. Funding: This study was funded by a Changi General Hospital Research project grant.
- Role of the funding body in design of the study, collection, analysis and interpretation of data and in writing the manuscript: none.
- Authors Contributions: DR, PSD, TTY, YCJQ, ATC, HDJ, WSR contributed equally to the design, collection of data and writing the paper. JM carried out the statistical analysis. All authors have read and approved the final manuscript.
- Acknowledgements: Geraldine Lim, Clinical trials and research unit, Changi General Hospital, Singapore.
References
- Norgren, Hiatt WR, Dormandy JA, et al. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). Journal of Vascular Surgery 45 (2007): S5-S67.
- Barrett BJ, Parfrey PS. Prevention of nephrotoxicity induced by radiocontrast agents. N Engl J Med 331 (1994): 1449-1450.
- Tepel M, Aspelin P, Lamiere N. Contrast-induced nephropathy: A clinical and evidence- based approach. Circulation 113 (2006): 1799-1806.
- Goldenberg I, Matetzky S. Nephropathy induced by contrast media: pathogenesis, risk factors and preventive strategies. CMAJ 172 (2005): 1461-1471.
- Paraskevas KI, Giannoukas AD, Mikhailidis DP. The impact of impaired renal function on long-term outcomes in patients with peripheral arterial disease. Angiology 61 (2010): 415- 416.
- Paraskevas KI, Giannoukas AD, Mikhailidis DP. Renal function impairment in peripheral arterial disease: an important parameter that should not be neglected. Ann Vasc Surg 23 (2009): 690-699.
- O’Hare AM, Bertenthal D, Shlipak MG, et al. Impact of renal insufficiency on mortality in advanced lower extremity peripheral arterial disease. J Am Soc Nephrol 16 (2005): 514-519.
- Zaraca F, Wiedermann CJ, Ebner H. Contrast media-induced nephropathy in patients undergoing angiography prior to or during vascular surgery: a systematic review. Minerva Chir 66 (2011): 553-560.
- Carbonell N, Blasco M, Sanjuan R, et al. Intravenous N-Acetylcysteine for preventing contrast-induced nephropathy: a randomised trial. Int J Cardiol 115 (2007): 57-62.
- Fikret Er, Nia AM, Henning D, et al. Ischaemic Preconditioning for prevention of contrast medium-induced nephropathy. Randomized Pilot RenPro Trial (Renal Protection Trial) Circulation 126 (2012): 296-303.
- World Medical Association Declaration of Helsinki Ethical Principles for Medical Research Involving Human Subjects. JAMA 310 (2013): 2191-2194.
- Grenz A, Osswald H, Eckle T, et al. The renovascular A2B adenosine receptor protects the kidney from ischemia. PLoS Med 5 (2008): e137.
- Whittaker P, Przyklenk K. Remote-Conditioning Ischemia Provides a Potential Approach to Mitigate Contrast Medium-Induced Reduction in Kidney Function: A Retrospective Observational Cohort Study. Cardiology 119 (2011): 145-150.
- Rudnick M, Feldman H. Contrast induced nephropathy: what are the true clinical consequences. Clin J Am Soc Nephrol 3 (2008): 263-272.
- KDIGO Clinical practice guidelines for AKI. Kid Inter Supp 2 (2012): 124-138.
- Darling CE, Solari PB, Smith CS, et al. Postconditioning’ the human heart: multiple balloon inflations during primary angioplasty may confer cardioprotection. Basic Res Cardiol 102 (2007): 274-278.
- Wang K, Duan CY, Wu J, et al. Predictive Value of Neutrophil Gelatinase-Associated Lipocalin for Contrast-Induced Acute Kidney Injury After Cardiac Catheterization: A Meta-analysis. Can J Cardiol 32 (2016): 1033.e19-1033.e29.
- Moledina DG, Hall IE, Thiessen-Philbrook H, et al. Performance of Serum Creatinine and Kidney Injury Biomarkers for Diagnosing Histologic Acute Tubular Injury. Am J Kidney Dis 70 (2017): 807-816.
- Navarese EP, Gurbel PA, Andreotti F, et al. Prevention of contrast induced acute kidney injury in patients undergoing cardiovascular procedures-a systematic review and network meta-analysis. Plos One 12 (2017): e0168726.
- Healy DA, Feeley I, Keogh CJ, et al. Remote ischemic conditioning and renal function after contrast- enhanced CT scan: A randomized trial. Clin Invest Med 38 (2015): E110-E118.
- Wong GTC, Lee EYP, Irwin NG. Contrast induced nephropathy in vascular surgery. British Journal of Anaesthesia 117 (2016): ii63–ii73.
- Menting TP, Sterenborg TB, Waal YD, et al. Remote Ischemic Preconditioning to Reduce Contrast-Induced Nephropathy: A Randomized Controlled Trial, European Journal of Vascular and Endovascular Surgery 50 (2015): 527- 532.
- Giacoppo D, Gargiulo G, Buccheri S, et.al. Preventive strategies for contrast-induced acute kidney injury in patients undergoing percutaneous coronary procedure: evidence from a hierarchical Bayesian network meta-analysis of 124 trials and 28,240 patients. Circ Cardiovasc Interv 10 (2017): e004383.
- Roy D, Pande SD, Aw TC, et al. Remote ischemic preconditioning in prevention of contrast induced nephropathy: A Randomised Controlled Trial (2019).


 Impact Factor: * 4.2
Impact Factor: * 4.2 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 