Retrospective Analysis of Early Stage Mucinous Ovarian Cancersystematic Lymphadenectomy May Be Omitted
Article Information
Ning Li1, Guangwen Yuan1, Xiaoguang Li1, Hongwen Yao1, Yan Song2, Lingying Wu1*
1Department of Gynecological Oncology, National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China
2Department of Pathology, National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China
*Corresponding Author: Lingying Wu, Department of Gynecological Oncology, National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, China
Received: 18 August 2019;Accepted: 13 September 2019;Published: 02 October 2019
Citation: Ning Li, Guangwen Yuan, Xiaoguang Li, Hongwen Yao, Yan Song, Lingying Wu. Retrospective Analysis of Early Stage Mucinous Ovarian Cancersystematic Lymphadenectomy May Be Omitted. Journal of Cancer Science and Clinical Therapeutics 3 (2019): 171-177.
View / Download Pdf Share at FacebookAbstract
Purpose: Mucinous ovarian cancer is a less common epithelial ovarian cancer. Mucinous early stage ovarian cancer (mEOC) seems less aggressive than other histologic types and require more conservative treatment. This analysis was conducted to explore the clinical outcome and appropriate treatment of mEOC.
Methods: Data was extracted from patients with stage I/II ovarian cancer from 1999 to 2010 at our institution. Patients were classified into mucinous group (n=45) and non-mucinous groups (n=159). Clinical features and survival outcomes were compared between the two groups.
Results: The 3-year/5-year progression free survival (PFS) of the mucinous were significantly longer than the non-mucinous group (96% /91.0% vs. 82.4% /75.1%, P=0.01). 40.0% of mEOC patients underwent systematic lymphadenectomy, and there was no significant differences of recurrence rate whether they received this procedure. 35.6% patients of mEOC underwent FSS and none of them experienced recurrence. 68.8% of mEOC patients received adjuvant chemotherapy.
Conclusion: mEOC has longer PFS than other histologic types. Systematic lymphadenectomy may be omitted in mEOC patients.
Keywords
Ovarian cancer; Mucinous adenocarcinoma; Lymphadenectomy; Recurrence
Ovarian cancer articles, Mucinous adenocarcinoma articles, Lymphadenectomy articles, Recurrence articles
Ovarian cancer articles Ovarian cancer Research articles Ovarian cancer review articles Ovarian cancer PubMed articles Ovarian cancer PubMed Central articles Ovarian cancer 2023 articles Ovarian cancer 2024 articles Ovarian cancer Scopus articles Ovarian cancer impact factor journals Ovarian cancer Scopus journals Ovarian cancer PubMed journals Ovarian cancer medical journals Ovarian cancer free journals Ovarian cancer best journals Ovarian cancer top journals Ovarian cancer free medical journals Ovarian cancer famous journals Ovarian cancer Google Scholar indexed journals Mucinous adenocarcinoma articles Mucinous adenocarcinoma Research articles Mucinous adenocarcinoma review articles Mucinous adenocarcinoma PubMed articles Mucinous adenocarcinoma PubMed Central articles Mucinous adenocarcinoma 2023 articles Mucinous adenocarcinoma 2024 articles Mucinous adenocarcinoma Scopus articles Mucinous adenocarcinoma impact factor journals Mucinous adenocarcinoma Scopus journals Mucinous adenocarcinoma PubMed journals Mucinous adenocarcinoma medical journals Mucinous adenocarcinoma free journals Mucinous adenocarcinoma best journals Mucinous adenocarcinoma top journals Mucinous adenocarcinoma free medical journals Mucinous adenocarcinoma famous journals Mucinous adenocarcinoma Google Scholar indexed journals Lymphadenectomy articles Lymphadenectomy Research articles Lymphadenectomy review articles Lymphadenectomy PubMed articles Lymphadenectomy PubMed Central articles Lymphadenectomy 2023 articles Lymphadenectomy 2024 articles Lymphadenectomy Scopus articles Lymphadenectomy impact factor journals Lymphadenectomy Scopus journals Lymphadenectomy PubMed journals Lymphadenectomy medical journals Lymphadenectomy free journals Lymphadenectomy best journals Lymphadenectomy top journals Lymphadenectomy free medical journals Lymphadenectomy famous journals Lymphadenectomy Google Scholar indexed journals Recurrence articles Recurrence Research articles Recurrence review articles Recurrence PubMed articles Recurrence PubMed Central articles Recurrence 2023 articles Recurrence 2024 articles Recurrence Scopus articles Recurrence impact factor journals Recurrence Scopus journals Recurrence PubMed journals Recurrence medical journals Recurrence free journals Recurrence best journals Recurrence top journals Recurrence free medical journals Recurrence famous journals Recurrence Google Scholar indexed journals ovarian carcinomas articles ovarian carcinomas Research articles ovarian carcinomas review articles ovarian carcinomas PubMed articles ovarian carcinomas PubMed Central articles ovarian carcinomas 2023 articles ovarian carcinomas 2024 articles ovarian carcinomas Scopus articles ovarian carcinomas impact factor journals ovarian carcinomas Scopus journals ovarian carcinomas PubMed journals ovarian carcinomas medical journals ovarian carcinomas free journals ovarian carcinomas best journals ovarian carcinomas top journals ovarian carcinomas free medical journals ovarian carcinomas famous journals ovarian carcinomas Google Scholar indexed journals mucinous carcinoma articles mucinous carcinoma Research articles mucinous carcinoma review articles mucinous carcinoma PubMed articles mucinous carcinoma PubMed Central articles mucinous carcinoma 2023 articles mucinous carcinoma 2024 articles mucinous carcinoma Scopus articles mucinous carcinoma impact factor journals mucinous carcinoma Scopus journals mucinous carcinoma PubMed journals mucinous carcinoma medical journals mucinous carcinoma free journals mucinous carcinoma best journals mucinous carcinoma top journals mucinous carcinoma free medical journals mucinous carcinoma famous journals mucinous carcinoma Google Scholar indexed journals chemotherapy articles chemotherapy Research articles chemotherapy review articles chemotherapy PubMed articles chemotherapy PubMed Central articles chemotherapy 2023 articles chemotherapy 2024 articles chemotherapy Scopus articles chemotherapy impact factor journals chemotherapy Scopus journals chemotherapy PubMed journals chemotherapy medical journals chemotherapy free journals chemotherapy best journals chemotherapy top journals chemotherapy free medical journals chemotherapy famous journals chemotherapy Google Scholar indexed journals lymphatic metastasis articles lymphatic metastasis Research articles lymphatic metastasis review articles lymphatic metastasis PubMed articles lymphatic metastasis PubMed Central articles lymphatic metastasis 2023 articles lymphatic metastasis 2024 articles lymphatic metastasis Scopus articles lymphatic metastasis impact factor journals lymphatic metastasis Scopus journals lymphatic metastasis PubMed journals lymphatic metastasis medical journals lymphatic metastasis free journals lymphatic metastasis best journals lymphatic metastasis top journals lymphatic metastasis free medical journals lymphatic metastasis famous journals lymphatic metastasis Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals Pathological slices articles Pathological slices Research articles Pathological slices review articles Pathological slices PubMed articles Pathological slices PubMed Central articles Pathological slices 2023 articles Pathological slices 2024 articles Pathological slices Scopus articles Pathological slices impact factor journals Pathological slices Scopus journals Pathological slices PubMed journals Pathological slices medical journals Pathological slices free journals Pathological slices best journals Pathological slices top journals Pathological slices free medical journals Pathological slices famous journals Pathological slices Google Scholar indexed journals
Article Details
1. Introduction
Growing evidence suggests that primary mucinous ovarian carcinomas have a distinct clinical features and course when compared to non-mucinons ovarian carcinomas [1, 2]. Patients with mucinous carcinoma of the ovary are often diagnosed with early-stage disease and have a good prognosis. One study reported a startling 87% of mucinous ovarian cancer confirmed at stage I [3]. The 5-year progression-free survival (FPS) is about 80% to 90% at early stage [4]. The majority of mEOCs are moderate or well-differentiated. mEOC is less aggressive and has a lower rate of lymphatic metastasis compared with other types [5].
Mucinous epithelial ovarian cancer is a separate entity requiring specific treatment. Some studies indicated that systematic lymphadenectomy may be omitted in early stage cases [6]. For adjuvant chemotherapy, some clinicians suggested the gastrointestinal regimens are appropriate because mucinous carcinomas of the ovary are similar to gastrointestinal tumors. The tailored treatment for mucinous ovarian cancer is still unknown because its rarity. We retrospectively analyzed all early stage (FIGO stage I-II) mucinous ovarian cancers over ten years in Chinese National Cancer Center/Cancer Hospital. The aims of this study were to provide the clinical and pathologic features and explore proper treatment of mEOC.
2. Materials and Methods
Medical charts of patients diagnosed with early stage ovarian cancer (EOC) from 1999 to 2010 at National Cancer Center/Cancer Hospital were reviewed. The data of patients with non-mucinous early stage ovarian cancer was also obtained for comparison. Pathological slices were reevaluated and restaged based on the 2014 International Federation of Gynecology and Obstetrics (FIGO) ovarian cancer staging criteria [7]. This study underwent appropriate IRB approval. Patients with borderline mucinous carcinoma and mucinous carcinomas metastatic to the ovary from other primary sites were excluded. No patients in final analysis were treated before the surgery. The time from surgery to recurrence or last disease-free follow-up time was calculated as progression free survival (PFS). Few early stage mucinous cancer patients died in our study; thus overall survival time was not calculated in our study.
The population was classified according to histologic diagnosis as the mucinous group and three non-mucinous groups (serous group, endometrioid group, and clear cell group). Continuous values were expressed by mean ± standard deviations or median value and were tested by t-tests or Mann-Whitney tests. Categorical variables were compared using Chi-square tests. PFS times were calculated by Kaplan-Meier analysis. We applied the log-rank test and cox regression between survival curves for univariate analysis and multivariate analysis respectively. A P value of less than 0.05 was considered statistically significant. Data were analyzed using SPSS 20.0 software.
3. Results
204 early stage ovarian cancer patients were included in the final analysis, in which mucinous subtype occupied 22.1% of the population. Patients were classified as mucinous group (n=45/22.1%) and other three control groups (n=159/77.9%). The most common pathologic type for non-mucinous group was serous adenocarcinoma (n=88/43.1%), followed by endometrioid carcinoma (n=37 /18.1%), then
clear cell carcinoma (n=34/16.7%). The early stage mucinous ovarian cancer showed different clinical and pathologic features compared with non-mucinous pathologic types. Mucinous ovarian cancer presented with larger proportion of stage I compared with a serous group (mucinous vs. serous, 93.4% vs. 47.7%, P < 0.01) and clear cell group (mucinous vs. serous, 93.4% vs. 82.4%, P < 0.01) (details of sub-stage distributions showed in table 1). The differentiation of mEOC was notably better (G1:84.4%, G2-2:15.6%) than the control groups’: serous (G1:25.0%, G2-3:75.0%, P<0.01), endometrioid (G1:24.3%, G2-3:75.7%, P<0.01) and clear cell(G1:29.4%, G2-3:70.6%, P<0.01). (Table 1.) 70.7% (29/41) of patients with mucinous cancers showed elevated abnormal CA12-5 levels but was significantly lower than serious type (serous vs. mucinous; mean/median, 652.4/225U/ml vs. 1865/77U/ml, P=0.02). 60.6% (20/33) of patients with mucinous tumors had elevated CA19-9, which was the highest values compared with a serous group (mucinous vs. serous; mean/median, 87825/79U/ml vs. mean/median, 138.9/14.3U/ml, P < 0.01) and clear cell group (mucinous vs. clear cell; mean/median, 87825/79U/ml vs. mean/median, mean/median, 86.5/22, P=0.02) (Table 1). The mEOC was significantly larger than the control groups (mucinous vs. serous, endometrioid and clear cell, 19.9 ± 9.8 cm vs. 12.4±7.2 cm, 11.4 ± 5.9 cm and 12.8 ± 6.1 cm; P < 0.01, P < 0.01 and P=0.01 respectively) (Table 1).
For primary surgical treatment, our study indicated that systematic pelvic lymphadenectomy may be omitted in mucinous ovarian cancer patients with tumors grossly confined to ovaries if suspicious lymph nodes were not detected during surgery. In our study, 40.0% mucinous patients received systematic lymphadenectomy and 60.0% patients only had lymphatic sampling or no lymph nodes dissection. Survival analysis could not be performed because only five patients experienced recurrence and one of them died. Chi-square analysis showed that there were no significant differences of recurrence rate between two groups. Fertility sparing surgery (FSS) may be considered in selected mucinous ovarian cancers. 35.6% patients of mucinous tumors underwent FSS and none of these patients experienced recurrence after at least 59-month follow-up (Table 2). 64.4% mEOC patients received postsurgical chemotherapy (paclitaxel plus carboplatin or cisplatin). None of patients opted observation after surgery relapse (Table 2).
aBetween groups comparisons were conducted by non-parameter test of tumor marker values; bG1: well-differentiation, G2-3: moderate-poor differentiation; cPTX: paclitaxel, CBP: carboplatin, DDP: cisplatin; dCourses of chemo: Courses of adjuvant chemotherapy
Table 1: Clinicopathological characteristics and treatment according to histological subtype.
|
|
|
Recurrence |
|
|
|
|
|
Yes |
No |
P |
|
Systematic lymphadenectomy |
Yes 18 No 27 |
4 1 |
14 26 |
0.63 |
|
Adjuvant Chemotherapy |
Yes 29 No 16 |
5 0 |
24 16 |
0.45 |
Table 2: Treatment and recurrence for early stage mucinous ovarian cancer.
The median follow-up period was 79 months, ranging from 4 months to 203 months. Five mEOC patients experienced recurrence and one of them died. The patient who died was recurrent in 8 months and resistant to platinum based chemotherapy. When compared the mucinous ovarian cancer and other pathologic subtypes, there were significant differences in of 3-year FPS (muscinous vs. non-mucinous: 96% (95% CI: 90.0- 99.9 vs. 82.4% (95% CI: 76.5- 88.3, P=0.01) and 5-year PFS (mucinous vs. non-mucinous: 91.0% (95%CI: 82.6-99.4) vs. 75.1% (95%CI: 68.2-82.0), P=0.01). Furthermore, the mucinous group showed significantly superior 3 year/ 5 year PFS than the serous group [mucinous vs. serous, 96% (95% CI: 90.0- 99.9) /91.0% (95%CI: 82.6-99.4) vs. 80.4% (95% CI: 72.0- 88.8) /73.2% (95%CI: 63.8-82.6, P=0.01) and the clear cell group [mucinous vs. clear cell, 96% (95% CI: 90.0- 99.9) /91.0% (95%CI: 82.6-99.4) vs 70.6% (95% CI: 55.3- 85.9) /67.6% (95%CI: 51.9-83.3, P=0.01) (Figure 1a-1c). However, univariate analysis and multivariate analysis failed to show the mucinous histology as an independent factor for PFS in early stage ovarian cancer. Instead, stage [stage II vs stage I, hazard ratio (HR)=3.2, 95% CI: 1.7-6.2, P=0.01], peritoneal biopsy (biopsy vs. non-biopsy, HR=0.27, 95%CI: 0.14-0.52, P < 0.01), and differentiation (Grade 2-3 vs. Grade 1, HR=2.3, 95%CI: 1.2-4.9, P=0.03) were independent prognostic factors for early stage ovarian cancer.
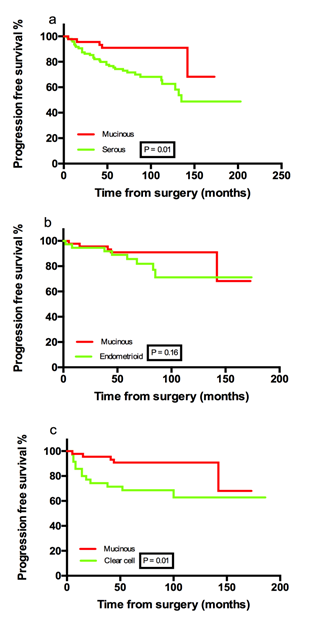
Figure 1: Progression free survival percentage.
4. Discussion
The studies on mEOC are limited because its rarity. It is showed that the mEOC had a low incidence, ranging from 3% to 5% in EOC population [3, 8]. Mucinous ovarian cancers tend to present at early stage. In a large scale central pathologic review, 70.3% of mEOCs were classified as stage I-II [2]. In our study, mEOC accounted for 20.3% early stage epithelial ovarian cancers. The 3-year/5-year PFS of patients with mucinous tumors in this study were 96% /91.0%. Because only 3 stage II mEOC patients were included, the calculated PFS rates reflected the prognosis of stage I mEOC rather than early-stage mEOC in our study. The result was consistent with a study involving 410 patients with stage I mEOC which reported the 5-year PFS was 90.8% [9]. In all, our studies demonstrated that early stage mucinous ovarian cancers enjoy pleasant prognosis; less aggressive treatment may be considered for these patients.
The value of systematic lymphadenectomy should be reconsidered for early stage mucinous ovarian cancers. Several studies reported that the prevalence of lymphatic metastasis in patients with mucinous tumors was excessively rare. A review include 14 studies showed that only 2.6% of stage I-II mucinous ovarian cancer had lymph node metastasis. Two studies involving 295 mEOCs, grossly noted to be stage I, found no lymph node involvement in all the 113 patients who underwent lymphadenectomy; the authors also showed that the lymphadenectomy did not improve prognosis [6, 10]. In our study, systematic pelvic lymph node dissection was performed in 40% mucinous ovarian cancer patients, and had no effect on recurrence rate. For 5 mucinous tumor patients experienced recurrence, only one did not receive lymph node dissection. Our study preliminarily supports the previous studies and challenges the necessity of lymphadenectomy in early stage mEOC. FSS seems a safe option for mucinous ovary cancer patients without obvious risk factors. In our study, 35.6% patients of mucinous tumors underwent FSS and none of these patients experienced recurrence after at least 59-month follow-up. Our institutional experience suggest that young patients with stage IA well-differentiated mucinous tumors may chose FSS and observation after surgery.
Adjuvant chemotherapy for Meoc is interesting. Despite a better prognosis at early stage, mEOC seems have worse PFS and OS compared with non-mucinous histologic types when it progresses into advanced-stage; most authors attributed this to more intensive chemo-resistance [11-13]. The chemo-resistance nature of mucinous ovarian cancer challenges the value of chemotherapy for early stage patients. In our study, only 14 stage IA/B mucinous patients were exempted from chemotherapy; these patients were all well-differentiated with or without completed staging surgery or FSS. After a minimum follow-up of 40 months, no recurrence was observed. It is implied that for stage I mucinous ovarian tumors without risk factors (IC, moderate/poor differentiation), even for those who did not receive completed staging surgery, observation after operation may be a safe option.
When stratified according to differentiation, the prognosis of mucinous and non-mucinous early-stage EOC was similar in our study. Several studies indicated that well-differentiation contributed to the good prognosis of mEOC. One study reported that tumor grade, not histology, independently predicted the relapse of stage I ovarian tumor [14]. Our study revealed similar results. Multivariate analysis showed that stage and differentiation, rather than histology, appeared to be independent prognostic factors in early stage ovarian cancers.
Our study also gave clues to why mEOC can be detected at an early stage. It is observed that mEOC tends to have a larger tumor volume with an intact capsule than other histologic types. The mean/median diameter of ovarian mass in our investigation was 19.9/18 cm, which was significantly larger than other types. The positive rate of ascites or peritoneal washing was also lower than the non-mucinous group in our analysis. It is reasonable to speculate that the mEOC patients may have more symptoms due to the large mass but, there is less of a chance for the cancer to spread.
5. Conclusion
In conclusion, most of mucinous ovarian cancers are diagnosed at an early stage with good pathologic features. The overall prognosis of early stage mEOC is better than non-mucinous counterparts. The treatment of early stage mEOC should be tailored. The extension of surgery and administration of adjuvant chemotherapy in a subset of early-stage mEOC patients should be explored by future studies.
References
- Soslow Robert A. Histologic subtypes of ovarian carcinoma: an overview. Int J Gynecol Pathol 27 (2008): 161-174.
- Shimada M, Kigawa J, Ohishi Y, et al. Clinicopathological characteristics of mucinous adenocarcinoma of the ovary. Gynecol Oncol 113 (2009): 331-334.
- Seidman JD, Horkayne-Szakaly I, Haiba M, et al. The histologic type and stage distribution of ovarian carcinomas of surface epithelial origin. Int J Gynecol Pathol 23 (2003): 41-44.
- Alexandre J, Ray-Coquard I, Selle F, et al. Mucinous advanced epithelial ovarian carcinoma: clinical presentation and sensitivity to platinum-paclitaxel-based chemotherapy, the GINECO experience. Ann Oncol 12 (2010): 2377-2381.
- Morice P, Joulie F, Camatte S, et al. Lymph node involvement in epithelial ovarian cancer: analysis of 276 pelvic and paraaortic lymphadenectomies and surgical implications. J Am Coll Surg 197 (2003): 198-205.
- Cho, Yun-Hyun, Dae-Yeon Kim, et al. Is complete surgical staging necessary in patients with stage I mucinous epithelial ovarian tumors?. Gynecol Oncol 03 (2006): 878-882.
- Prat Jaime. Staging classification for cancer of the ovary, fallopian tube, and peritoneum. Int J Gynecol Obstet 124 (2014): 1-5.
- Seidman JD, Kurman RJ, Ronnett BM. Primary and metastatic mucinous adenocarcinomas in the ovaries: incidence in routine practice with a new approach to improve intraoperative diagnosis Am J Surg Pathol 27 (2003): 985-993.
- Vergote I, De Brabanter J, Fyles A, et al. Prognostic importance of degree of differentiation and cyst rupture in stage I invasive epithelial ovarian carcinoma. The lancet 357 (2001): 176-182.
- Schmeler KM, et al. Prevalence of lymph node metastasis in primary mucinous carcinoma of the ovary. Obstet Gynecol, 116 (2010): 269-273.
- Hess V, A'Hern R, Nasiri N. Mucinous epithelial ovarian cancer: a separate entity requiring specific treatment. J Clin Oncol 22 (2004): 1040-1044.
- Pectasides D, Fountzilas G, Aravantinos G, et al. Advanced stage mucinous epithelial ovarian cancer: the Hellenic Cooperative Oncology Group experience. Gynecol Oncol 97 (2005): 436-441.
- Winter WE, Maxwell GL, Tian C, et al. Prognostic factors for stage III epithelial ovarian cancer: a Gynecologic Oncology Group Study. Clin Oncol 25 (2007): 3621-3627.
- Rodríguez IM, Jaime P. Mucinous tumors of the ovary: a clinicopathologic analysis of 75 borderline tumors (of intestinal type) and carcinomas. Am J Surg Pathol 26 (2002): 139-152.


 Impact Factor: * 4.1
Impact Factor: * 4.1 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 