Trajectories of Hospitalization in three Groups of Patients with Incurable Gastric Cancer Identified by Hierarchical Clustering: A Retrospective Analysis
Article Information
Yasuko Murakawa*, Masato Sakayori, and Kazunori Otsuka
Cancer Chemotherapy Department, Miyagi Cancer Center, Natori, Japan
*Corresponding Author: Dr. Yasuko Murakawa, Cancer Chemotherapy Department, Miyagi Cancer Center, Nodayama 47-1, Medeshima, Natori, Japan
Received: 05 October 2020; Accepted: 12 October 2020; Published: 27 October 2020
Citation: Yasuko Murakawa, Masato Sakayori, and Kazunori Otsuka. Trajectories of Hospitalization in three Groups of Patients with Incurable Gastric Cancer Identified by Hierarchical Clustering: A Retrospective Analysis. Journal of Cancer Science and Clinical Therapeutics 4 (2020): 499-510.
View / Download Pdf Share at FacebookAbstract
Purpose: The therapeutic goal for advanced solid malignancies is not to achieve cure but to prolong survival and maintain quality of life (QOL). To date, no study has reported the trajectory of the QOL throughout the clinical course of a patient with advanced malignancy. As hospitalization is considered a predictor of QOL, we retrospectively analyzed the trajectory of hospitalization in patients with incurable gastric cancer throughout the clinical course.
Methods: The data of 85 patients with incurable gastric cancer were collected, including age, sex, Eastern Cooperative Oncology Group (ECOG) Performance Status (PS), treatment, histology, sites of metastases at first consultation, planned and unplanned hospitalization throughout the clinical course, and overall survival (OS). We ranked the patients by OS and hospitalization using a hierarchical clustering analysis.
Results: Three clusters were identified corresponding to short, intermediate, and long OS/hospitalization (Clusters 1, 2, and 3, respectively). Patients in Cluster 3 were more likely to have an ECOG PS of 0-2 and receive palliative chemotherapy than the other clusters. No other differences in histology, age, sex, and or extra-peritoneal metastasis sites were observed between the three groups. In Cluster 3, planned hospitalization accumulated gradually during the early clinical phase, while unplanned hospitalization accumulated rapidly in later phases.
Conclusions: No specific characteristics were associated with short, intermediate, and long OS/hospitalization. Patients in the long OS/hospitalization group exhibited a rapid accumulation of unplanned hospitalization during the latter clinical course. Further research is needed to identify specific predictors of and measures to avoid a long OS/hospitalization.
Keywords
Incurable gastric cancer; Overall survival; Quality of life; Hospitalization; Planned hospitalization; Unplanned hospitalization
Incurable gastric cancer articles; Overall survival articles; Quality of life articles; Hospitalization articles; Planned hospitalization articles; Unplanned hospitalization articles
Incurable gastric cancer articles Incurable gastric cancer Research articles Incurable gastric cancer review articles Incurable gastric cancer PubMed articles Incurable gastric cancer PubMed Central articles Incurable gastric cancer 2023 articles Incurable gastric cancer 2024 articles Incurable gastric cancer Scopus articles Incurable gastric cancer impact factor journals Incurable gastric cancer Scopus journals Incurable gastric cancer PubMed journals Incurable gastric cancer medical journals Incurable gastric cancer free journals Incurable gastric cancer best journals Incurable gastric cancer top journals Incurable gastric cancer free medical journals Incurable gastric cancer famous journals Incurable gastric cancer Google Scholar indexed journals Overall survival articles Overall survival Research articles Overall survival review articles Overall survival PubMed articles Overall survival PubMed Central articles Overall survival 2023 articles Overall survival 2024 articles Overall survival Scopus articles Overall survival impact factor journals Overall survival Scopus journals Overall survival PubMed journals Overall survival medical journals Overall survival free journals Overall survival best journals Overall survival top journals Overall survival free medical journals Overall survival famous journals Overall survival Google Scholar indexed journals Quality of life articles Quality of life Research articles Quality of life review articles Quality of life PubMed articles Quality of life PubMed Central articles Quality of life 2023 articles Quality of life 2024 articles Quality of life Scopus articles Quality of life impact factor journals Quality of life Scopus journals Quality of life PubMed journals Quality of life medical journals Quality of life free journals Quality of life best journals Quality of life top journals Quality of life free medical journals Quality of life famous journals Quality of life Google Scholar indexed journals Hospitalization articles Hospitalization Research articles Hospitalization review articles Hospitalization PubMed articles Hospitalization PubMed Central articles Hospitalization 2023 articles Hospitalization 2024 articles Hospitalization Scopus articles Hospitalization impact factor journals Hospitalization Scopus journals Hospitalization PubMed journals Hospitalization medical journals Hospitalization free journals Hospitalization best journals Hospitalization top journals Hospitalization free medical journals Hospitalization famous journals Hospitalization Google Scholar indexed journals Planned hospitalization articles Planned hospitalization Research articles Planned hospitalization review articles Planned hospitalization PubMed articles Planned hospitalization PubMed Central articles Planned hospitalization 2023 articles Planned hospitalization 2024 articles Planned hospitalization Scopus articles Planned hospitalization impact factor journals Planned hospitalization Scopus journals Planned hospitalization PubMed journals Planned hospitalization medical journals Planned hospitalization free journals Planned hospitalization best journals Planned hospitalization top journals Planned hospitalization free medical journals Planned hospitalization famous journals Planned hospitalization Google Scholar indexed journals Unplanned hospitalization articles Unplanned hospitalization Research articles Unplanned hospitalization review articles Unplanned hospitalization PubMed articles Unplanned hospitalization PubMed Central articles Unplanned hospitalization 2023 articles Unplanned hospitalization 2024 articles Unplanned hospitalization Scopus articles Unplanned hospitalization impact factor journals Unplanned hospitalization Scopus journals Unplanned hospitalization PubMed journals Unplanned hospitalization medical journals Unplanned hospitalization free journals Unplanned hospitalization best journals Unplanned hospitalization top journals Unplanned hospitalization free medical journals Unplanned hospitalization famous journals Unplanned hospitalization Google Scholar indexed journals solid malignancy articles solid malignancy Research articles solid malignancy review articles solid malignancy PubMed articles solid malignancy PubMed Central articles solid malignancy 2023 articles solid malignancy 2024 articles solid malignancy Scopus articles solid malignancy impact factor journals solid malignancy Scopus journals solid malignancy PubMed journals solid malignancy medical journals solid malignancy free journals solid malignancy best journals solid malignancy top journals solid malignancy free medical journals solid malignancy famous journals solid malignancy Google Scholar indexed journals incurable cancer articles incurable cancer Research articles incurable cancer review articles incurable cancer PubMed articles incurable cancer PubMed Central articles incurable cancer 2023 articles incurable cancer 2024 articles incurable cancer Scopus articles incurable cancer impact factor journals incurable cancer Scopus journals incurable cancer PubMed journals incurable cancer medical journals incurable cancer free journals incurable cancer best journals incurable cancer top journals incurable cancer free medical journals incurable cancer famous journals incurable cancer Google Scholar indexed journals chemotherapy articles chemotherapy Research articles chemotherapy review articles chemotherapy PubMed articles chemotherapy PubMed Central articles chemotherapy 2023 articles chemotherapy 2024 articles chemotherapy Scopus articles chemotherapy impact factor journals chemotherapy Scopus journals chemotherapy PubMed journals chemotherapy medical journals chemotherapy free journals chemotherapy best journals chemotherapy top journals chemotherapy free medical journals chemotherapy famous journals chemotherapy Google Scholar indexed journals Oncology articles Oncology Research articles Oncology review articles Oncology PubMed articles Oncology PubMed Central articles Oncology 2023 articles Oncology 2024 articles Oncology Scopus articles Oncology impact factor journals Oncology Scopus journals Oncology PubMed journals Oncology medical journals Oncology free journals Oncology best journals Oncology top journals Oncology free medical journals Oncology famous journals Oncology Google Scholar indexed journals
Article Details
1. Introduction
The therapeutic goal for a patient with an advanced solid malignancy is to prolong survival and maintain the quality of life (QOL), rather than to achieve a curative outcome [1]. Although many studies have reported the overall survival (OS) outcomes of patients with incurable cancer, changes in the QOL throughout the clinical course have not been evaluated. Eastern Cooperative Oncology Group (ECOG) performance status (PS) and palliative chemotherapy status are generally considered prognostic factors for OS in patients with advanced solid malignancies. Several studies have shown that palliative chemotherapy generally does not prolong survival in patients with solid malignancies who have a poor ECOG PS [2, 3]. The American Society of Clinical Oncology advocates withholding palliative chemotherapy from patients with solid tumors and an ECOG PS of 3-4, and instead recommends best supportive care (BSC) [4]. However, in our previous study [5], almost half of patients with an ECOG PS of 3-4 selected palliative chemotherapy, while patients with an ECOG PS of 0-2 did not experience prolonged survival with palliative chemotherapy when compared to patients with an ECOG PS of 3-4.
Typically, QOL is evaluated using a questionnaire, and many studies that address changes in QOL are limited to specific phases of the clinical course. However, hospitalization is considered a predictor of QOL [6]. This variable can be divided into planned and unplanned hospitalization, of which the latter is generally considered unfavorable for patients. To date, no study has reported the trajectory of hospitalization throughout the clinical course. Gastric cancer was the fifth most common malignancy and the third leading cause of cancer-related mortality in 2018, according to Global Cancer Statistics [7]. Gastric cancer is even more common in Japan, with the second highest incidence and third highest mortality rate of all cancers according to a short-term projection method applied by the Projected Cancer Statistics 2018 [8]. Therefore, the first aim of this study was to classify patients with incurable gastric cancer, according to OS and hospitalization and clarify the characteristics of the resulting groups. The second aim was to analyze the trajectory of hospitalization throughout the clinical course.
2. Methods
2.1 Patients
We retrospectively evaluated 85 patients with incurable gastric cancer who attended the Miyagi Cancer Center (Natori, Japan) between May 2014 and February 2018 and died up to May 2019. This study collected the following data: age, sex, ECOG PS, treatment (palliative chemotherapy vs. BSC), histology, sites of metastases at first consultation, OS, hospitalization (planned and unplanned), and the trajectory of hospitalization. Histologically, patients were divided into two types: intestinal type or diffuse type. The mixed type, which included both the intestinal and diffuse types, was categorized as the latter. The sites of metastases were the peritoneum, liver, lymph nodes, bone, anastomotic site/remaining stomach, lung/pleura, adrenal gland, others (ovary, skin, and brain), and locally advanced disease. The oncologist explained the benefits and limitations of palliative chemotherapy and BSC to all patients during their first consultation.
2.2 Statistical analyses
We used a hierarchical clustering analysis method to classify the patients based on OS and hospitalization. We then used the square of the Euclidean distance dissimilarity coefficient to determine the similarities or differences between patients. The results of the clustering analysis are presented in a dendrogram chart. The ECOG PS, treatment, histology, age, sex, and sites of metastases in the groups were compared using the chi-square test. A two-tailed P-value of <0.05 was considered significant. OS curves were estimated using the Kaplan-Meier method, and comparisons of survival between clusters were performed using the log-rank test. All statistical analyses were performed using the Statistical Package for the Social Science for Windows (version 24; SPSS Inc., Chicago, IL, USA).
3. Results
The cluster analysis with OS and hospitalization yielded three groups of patients (Figure 1) which are depicted in a scatter plot (Figure 2). The Kaplan-Meier analysis revealed that Cluster 3 had the longest OS of the three clusters (median OS: Cluster 3 vs. Cluster 1 and Cluster 2; 33.2 vs. 3.7 and 13.2 months, respectively; P<0.001 for both). Cluster 2 had a longer OS than Cluster 1 (median OS: 13.2 vs. 3.7 months; P<0.001; Figure 3). Cluster 3 also had the longest hospitalization (mean hospitalization: Cluster 3 vs. Cluster 1 and Cluster 2; 4.9 vs. 1.8 and 2.6 months, respectively; P<0.001 for both), and Cluster 2 had a longer hospitalization than Cluster 1 (mean hospitalization: 2.6 vs. 1.8 months; P<0.05). Consequently, Clusters 1, 2, and 3 were characterized as the short, intermediate, and long OS/hospitalization groups, respectively (Figure 4).
An analysis of the clinical characteristics revealed that more patients in Cluster 3 had an ECOG PS of 0-2 compared to those in Clusters 1 and 2 (ECOG PS 0-2/3-4: Cluster 1, 25/13; Cluster 2, 32/6; Cluster 3, 9/0; P<0.05). Patients in Cluster 3 were also more likely to have received palliative chemotherapy (palliative chemotherapy/BSC: Cluster 1, 23/15; Cluster 2, 35/3; Cluster 3, 9/0; P<0.01). Patients in Cluster 2 were less likely to present with metastasis in the peritoneum (metastasis in peritoneum, +/-: Cluster 1, 22/16; Cluster 2, 11/27; Cluster 3, 5/4; P<0.05). There were no differences in histology, age, sex, or other sites of metastases between the three groups (Table 1).
Next, we analyzed planned and unplanned hospitalization in the three groups (Figure 4). Here, planned hospitalization referred to a short-term stay for palliative chemotherapy, while unplanned hospitalization involved cancer-related symptoms, that usually needed a long-term stay. Cluster 3 had a longer duration of planned hospitalization than Clusters 1 and 2 (mean: 2.2 vs. 0.3 and 1.0 months, respectively; P<0.001 for both), while Cluster 2 had a longer duration than Cluster 1 (mean: 1.0 vs. 0.3 months; P<0.001). Cluster 3 also had a longer duration of unplanned hospitalization than Clusters 1 and 2 (mean: 2.7 vs. 1.5 and 1.6 months, respectively; P<0.05 for both), whereas there was no significant difference between Clusters 1 and 2. The trajectory of hospitalization was then analyzed in each group (Figure 5).
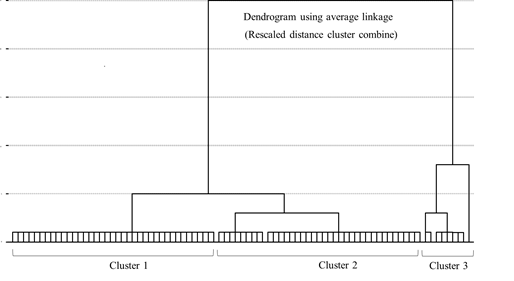
Figure 1: Dendrogram of patients with incurable gastric cancer categorized by overall survival and hospitalization.
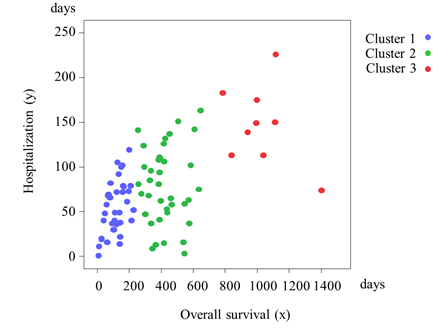
Figure 2: Correlation between overall survival and hospitalization in patients with incurable gastric cancer.
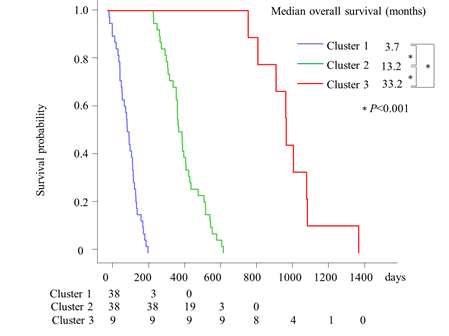
Figure 3: Kaplan-Meier curves of estimated overall survival in three groups of patients with incurable gastric cancer.
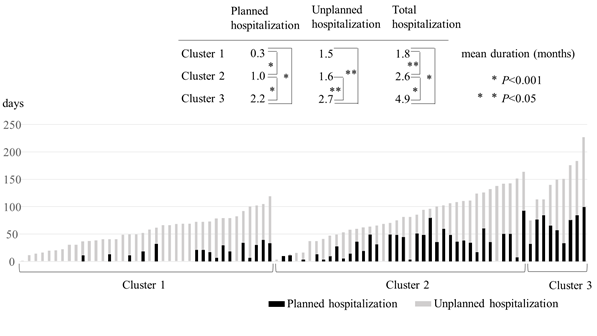
Figure 4: Planned and unplanned hospitalization in three groups of patients with incurable gastric cancer.
|
Variable |
Total 85 |
Cluster 1a 38 |
Cluster 2 38 |
Cluster 3 9 |
P-value |
|
ECOG PS |
|||||
|
0-2 |
66 (77.6%) |
25 (65.8%) |
32 (84.2%) |
9 (100%) |
<0.05* |
|
3-4 |
19 (22.4%) |
13 (34.2%) |
6 (15.8%) |
0 (0%) |
|
|
Treatment |
|||||
|
Palliative chemotherapy |
67 (78.8%) |
23 (60.5%) |
35 (92.1%) |
9 (100%) |
<0.01* |
|
BSC |
18 (21.2%) |
15 (39.5%) |
3 (7.9%) |
0 (0%) |
|
|
Histology |
|||||
|
Intestinal type |
23 (27.1%) |
10 (26.3%) |
8 (21.1%) |
5 (55.6%) |
0.110 |
|
Diffuse type |
62 (72.9%) |
28 (73.7%) |
30 (78.9%) |
4 (44.4%) |
|
|
Age (years) |
|||||
|
≥75 |
21 (24.7%) |
11 (28.9%) |
9 (23.7%) |
1 (11.1%) |
0.527 |
|
<75 |
64 (75.3%) |
27 (70.2%) |
29 (76.3%) |
8 (88.9%) |
|
|
Sex |
|||||
|
Female |
26 (30.6%) |
10 (38.5%) |
13 (34.2%) |
3 (33.3%) |
0.743 |
|
Male |
59 (69.4%) |
28 (73.7%) |
25 (65.8%) |
6 (66.7%) |
|
|
Site of metastasis Peritoneum |
|||||
|
(+) |
38 (44.7%) |
22 (57.9%) |
11 (28.9%) |
5 (55.6%) |
<0.05* |
|
(-) |
47 (55.3%) |
16 (42.1%) |
27 (71.1%) |
4 (44.4%) |
|
|
Liver |
|||||
|
(+) |
29 (34.1%) |
15 (39.5%) |
11 (28.9%) |
3 (33.3%) |
0.625 |
|
(-) |
56 (65.9%) |
23 (60.5%) |
27 (71.1%) |
6 (66.7%) |
|
|
Lymph node |
|||||
|
(+) |
57 (67.1%) |
27 (71.1%) |
23 (60.5%) |
7 (61.2%) |
0.478 |
|
(-) |
28 (32.9%) |
11 (28.9%) |
15 (39.5%) |
2 (38.9%) |
|
|
Locally advanced |
|||||
|
(+) |
20 (23.5%) |
7 (18.4%) |
10 (26.3%) |
3 (33.3%) |
0.550 |
|
(-) |
65 (76.5%) |
31 (81.6%) |
28 (73.7%) |
6 (66.7%) |
|
|
Bone |
|||||
|
(+) |
7 (8.2%) |
4 (10.5%) |
3 (7.9%) |
0 (0%) |
0.583 |
|
(-) |
78 (91.8%) |
34 (89.5%) |
35 (92.1%) |
9 (100%) |
|
|
Anastomotic site/remaining stomach |
|||||
|
(+) |
5 (5.9%) |
4 (10.5%) |
1 (2.6%) |
0 (0%) |
0.251 |
|
(-) |
80 (94.1%) |
34 (89.5%) |
37 (97.4%) |
9 (100%) |
|
|
Lung/pleura |
|||||
|
(+) |
5 (5.9%) |
2 (5.3%) |
3 (7.9%) |
0 (0%) |
0.648 |
|
(-) |
80 (94.1%) |
36 (94.7%) |
35 (92.1%) |
9 (100%) |
|
|
Adrenal gland |
|||||
|
(+) |
4 (4.7%) |
2 (5.3%) |
2 (5.3%) |
0 (0%) |
0.780 |
|
(-) |
81 (95.3%) |
36 (94.7%) |
36 (94.7%) |
9 (100%) |
|
|
Others |
|||||
|
(+) |
4 (4.7%) |
4 (10.5%) |
0 (0%) |
0 (0%) |
0.075 |
|
(-) |
81 (95.3%) |
34 (89.5%) |
38 (100%) |
9 (100%) |
|
*P<0.05
aClusters 1, 2, and 3 were characterized as the short, intermediate, and long OS/hospitalization groups, respectively.
Abbreviations: BSC-Best supportive care; ECOG-Eastern Cooperative Oncology Group; PS-Performance status
Table 1: Characteristics of the three groups clustered by overall survival and hospitalization duration.
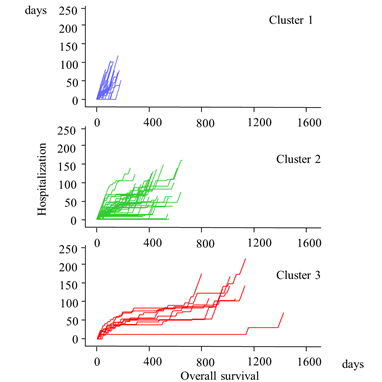
Figure 5: Trajectories of hospitalization vs. overall survival in three groups of patients with incurable gastric cancer.
Cluster 3 exhibited a gradual accumulation of planned hospitalization during the early phase, followed by a rapid accumulation of unplanned hospitalization. Cluster 1 experienced almost entirely unplanned hospitalization. Cluster 2 exhibited an intermediate pattern between those of Clusters 1 and 3.
4. Discussion
ECOG PS and palliative chemotherapy have been reported as independent prognostic factors for OS [9, 10]. Specifically, patients with a good ECOG PS are recommended to undergo palliative chemotherapy [11], and a meta-analysis of randomized controlled trials of advanced gastric cancer reported that chemotherapy extended OS by 6.7 months relative to BSC [12]. In this study, we accordingly classified patients into clusters and determined that patients in Cluster 3 were most likely to have an ECOG PS of 0-2 and to have received palliative chemotherapy compared to the other clusters. We noted that metastases of the peritoneum, lymph nodes, bone, lung, ovary, and brain have also been identified as potential prognostic factors for OS [13-17]. In this study, only the likelihood of peritoneal metastasis differed, with a higher incidence in Cluster 2 relative to the other clusters. These results might be attributable to the use of data collected at the first consultation which did not account for newly developed metastases.
Lauren’s histological classification classifies gastric cancers into two histological subtypes, intestinal and diffuse, as this variable has been reported to predict survival and responses to chemotherapy [18, 19]. In this study, however, we did not observe a significant association of either type with a long OS. We noted that the histological diagnoses in our cases were generally made from biopsy rather than surgical samples. Therefore, the diagnoses might not have reflected the true nature of the disease. In contrast to histology, age is not generally considered a prognostic factor in patients with advanced gastric cancer, and several studies reported that palliative chemotherapy is equally tolerable and effective in older and younger patients [20, 21]. Consistent with those observations, we did not observe an association of age with a long OS.
QOL has become increasingly important as the number of newly diagnosed patients with cancer continues to increase. Over time, improvements in the management of certain chemotherapy-associated toxicities have led to a shift from issues of physical QOL to issues of psychosocial QOL [22]. The most widely used measures of cancer-specific health-related QOL are the European Organization for Research and Treatment Quality of Life Questionnaire, version 3.0 (EORTC-QLQ-C30), McGill QOL questionnaire, and the EuroQol-5D (EQ-5D) [23]. These questionnaires have been used to investigate the trajectory of QOL among advanced-stage cancer patients in several studies [24, 25]. However, the physical and/or mental condition of the patient may make it difficult to administer a questionnaire, particularly if the patient has a poor ECOG PS and has reached the end-of-life stage. Moreover, many studies of the changes in QOL experienced by patients with incurable cancer patients are limited to specific clinical course phases, such as limited cycles of chemotherapy or phase III chemotherapy trials [26, 27].
Hospitalization has negative effects on the QOL. However, this option is needed in many situations. Planned hospitalization is necessary for the administration of palliative chemotherapy, radiation therapy, and surgery. In contrast, unplanned hospitalization is generally needed to treat chemotherapy-related toxicities and cancer-related symptoms and is especially undesirable for patients [28, 29]. Patients with end-stage disease exhibit a significant loss of QOL during hospitalization [30]. Notably, one previous study suggested the importance of an optimal discharge-planning system and early referral to palliative care to prevent hospital readmission [31].
The Japanese healthcare system features a unique combination of characteristics that have led to the overuse of tests and drugs, as well as relatively longer hospital stays than those in other countries [32]. One study reported that 27% of older patients in Japan experienced ≥90 days of hospitalization during their last year of life [33]. For patients with incurable malignancies, decision making regarding treatment is complex; therefore, oncologists need to assist the patients and their families [34]. In decision making, not only OS but the trajectory of QOL throughout the clinical course might be helpful.
In this study, a long OS was shown to correlate strongly with hospitalization. However, the trajectory of hospitalization tended to accumulate rapidly due to unplanned hospitalization during the last phase of the clinical course.
The major strength of this study was the classification of patients with incurable gastric cancer into three groups by OS/hospitalization as well as our analysis of various characteristics of these groups, including planned and unplanned hospitalization.
This study has several limitations. We used planned/unplanned hospitalization as a predictor of QOL; however, other factors are also associated with psychosocial QOL. In this study, predicting the length of OS/hospitalization was challenging. In the future, predicting OS and the trajectory of QOL throughout the clinical course is essential.
5. Conclusion
An ECOG PS of 0-2 and palliative chemotherapy were significantly associated with a long OS and hospitalization duration in our analysis. However, these factors were not specific to a particular group. Patients in the long OS/hospitalization group tended to accumulate unplanned hospitalization during the last phase of the clinical course. Further research is needed to identify the specific factors predictive of a long OS/hospitalization and the measures needed to avoid a long unplanned hospitalization.
Availability of data and materials
The raw data used in this study have been shared in Figshare (DOI: 10.6084/m9.figshare.10278155.)
Compliance with ethical standards
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Miyagi Cancer Center, reference number: No. 4, March 13, 2019) and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
According to the local ethics policy for the retrospective analysis of anonymized in-house clinical data, information about this study and a means of opting out were posted on a website.
Consent for Publication
Not applicable
Competing Interests
The authors declare that they have no competing interests.
References
- Browner I, Carducci MA. Palliative chemotherapy: Historical perspective, applications, and controversies. Semin Oncol 32 (2005): 145-155.
- Wheatley-Price P, Ali M, Balchin K, et al. The role of palliative chemotherapy in hospitalized patients. Curr Oncol 21 (2014): 187-192.
- Crosara Teixeira M, Marques DF, Ferrari AC, et al. The effects of palliative chemotherapy in metastatic colorectal cancer patients with an ECOG performance status of 3 and 4. Clin Colorectal Cancer 14 (2015): 52-57.
- Schnipper LE, Smith TJ, Raghavan D, et al. American Society of Clinical Oncology identifies five key opportunities to improve care and reduce costs: The top five list for oncology. J Clin Oncol 30 (2012): 1715-1724.
- Murakawa Y, Sakayori M, Otsuka K. Impact of palliative chemotherapy and best supportive care on overall survival and length of hospitalization in patients with incurable Cancer: A 4-year single institution experience in Japan. BMC Palliat Care 18 (2019): 45.
- Prince RM, Powis M, Zer A, et al. Hospitalisations and emergency department visits in cancer patients receiving systemic therapy: Systematic review and meta-analysis. Eur J Cancer Care (Engl) 28 (219): 12909.
- Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68 (2018): 394-424.
- Katanoda K, Kamo K, Saika K, et al. Short-term projection of cancer incidence in Japan using an age-period interaction model with spline smoothing. Jpn J Clin Oncol 44 (2014): 36-41.
- Fuchs CS, Muro K, Tomasek J, et al. Prognostic factor analysis of overall survival in gastric cancer from two phase III studies of second-line ramucirumab (REGARD and RAINBOW) using pooled patient data. J Gastric Cancer 17 (2017): 132-144.
- Zeng WJ, Hu WQ, Wang LW, et al. Long term follow up and retrospective study on 533 gastric cancer cases. BMC Surg 14 (2014): 29.
- Peppercorn JM, Smith TJ, Helft PR, et al. American society of clinical oncology statement: Toward individualized care for patients with advanced cancer. J Clin Oncol 29 (2011): 755-760.
- Wagner AD, Grothe W, Behl S, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev 8 (2017): CD004064.
- Mizrak Kaya D, Nogueras-González GM, Harada K, et al. Risk of peritoneal metastases in patients who had negative peritoneal staging and received therapy for localized gastric adenocarcinoma. J Surg Oncol 117 (2018): 678-684.
- Deng J, Sun D, Pan Y, et al. Ratio between negative and positive lymph nodes is suitable for evaluation the prognosis of gastric cancer patients with positive node metastasis. PLoS One 7 (2012): e43925.
- Koo DH, Ryu MH, Ryoo BY, et al. Improving trends in survival of patients who receive chemotherapy for metastatic or recurrent gastric cancer: 12 years of experience at a single institution. Gastric Cancer 18 (2015): 346-353.
- Uyeturk U, Arslan SH, Bal O, et al. Isolated ovarian metastasis of gastric cancer: Krukenberg tumor. Contemp Oncol (Pozn) 17 (2013): 515-519.
- Kraszkiewicz M, Wydmanski J. Brain metastases from stomach cancer - The role of different treatment modalities and efficacy of palliative radiotherapy. Rep Pract Oncol Radiother 20 (2014): 32-37.
- Jimenez Fonseca P, Carmona-Bayonas A, Hernández R, et al. Lauren subtypes of advanced gastric cancer influence survival and response to chemotherapy: Real-world data from the AGAMENON National Cancer Registry. Br J Cancer 117 (2017): 775-782.
- Ma J, Shen H, Kapesa L, et al. Lauren classification and individualized chemotherapy in gastric cancer. Oncol Lett 11 (2016): 2959-2964.
- Berger AK, Zschaebitz S, Komander C, et al. Palliative chemotherapy for gastroesophageal cancer in old and very old patients: A retrospective cohort study at the National Center for Tumor Diseases, Heidelberg. World J Gastroenterol 21 (2015): 4911-4918.
- Kim ST, Park KH, Oh SC, et al. Is chemotherapy in elderly patients with metastatic or recurrent gastric cancer as tolerable and effective as in younger patients? Asia Pac J Clin Oncol 8 (2012): 194-200.
- Carelle N, Piotto E, Bellanger A, et al. Changing patient perceptions of the side effects of cancer chemotherapy. Cancer 95 (2002): 155-163.
- Davis MP, Hui D. Quality of life in palliative care. Expert Rev Qual Life Cancer Care 2 (2017): 293-302.
- Giesinger JM, Wintner LM, Oberguggenberger AS, et al. Quality of life trajectory in patients with advanced cancer during the last year of life. J Palliat Med 14 (2011): 904-912.
- Kypriotakis G, Vidrine DJ, Francis LE, et al. The longitudinal relationship between quality of life and survival in advanced stage cancer. Psychooncology 25 (2016): 225-231.
- Tantoy IY, Cooper BA, Dhruva A, et al. Quality of life of patients with gastrointestinal cancers undergoing chemotherapy. Qual Life Res 27 (2018): 1865-1876.
- Hamaker ME, Schulkes KJ, Ten Bokkel Huinink D, et al. Evaluation and reporting of quality of life outcomes in phase III chemotherapy trials for poor prognosis malignancies. Qual Life Res 26 (2017): 65-71.
- Weaver C, Schiech L, Held-Warmkessel J, et al. Risk for unplanned hospital readmission of patients with cancer: Results of a retrospective medical record review. Oncol Nurs Forum 33 (2006): 44-52.
- Whitney RL, Bell JF, Tancredi DJ, et al. Unplanned hospitalization among individuals with cancer in the year after diagnosis. J Oncol Pract 15 (2019): 20-29.
- Buzgová R, Sikorová L, Kozáková R, et al. Predictors of change in quality of life in patients with end-stage disease during hospitalization. J Palliat Care 32 (2017): 69-76.
- Granda-Cameron C, Behta M, Hovinga M, et al. Risk factors associated with unplanned hospital readmissions in adults with cancer. Oncol Nurs Forum 42 (2015): 257-268.
- Nomura H, Nakayama T. The Japanese healthcare system. BMJ 331 (2005): 648-649.
- Ishizaki T, Shimmei M, Fukuda H, et al. Cumulative number of hospital bed days among older adults in the last year of life: A retrospective cohort study. Geriatr Gerontol Int 17 (2017): 737-743.
- Bakitas M, Kryworuchko J, Matlock DD, et al. Palliative medicine and decision science: The critical need for a shared agenda to foster informed patient choice in serious illness. J Palliat Med 14 (2011): 1109-1116.


 Impact Factor: * 4.1
Impact Factor: * 4.1 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 